pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed

pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed
Jungsoo Lee,Jisang Yun,Sung-Min Park,Kihyuk Shin,Hoon-Soo Kim,Byung-Soo Kim,Moon-Bum Kim,Hyun-Chang Ko
10.17966/JMI.2022.27.2.25 Epub 2022 July 01
Abstract
Background: Dermatologic diseases are generally managed at an outpatient clinic and most cases have very low mortality rates. However, some dermatologic diseases are life-threatening and only a few studies have assessed their mortality rates and associated factors.
Objective: To investigate the mortality of inpatients with dermatological diseases.
Methods: This retrospective study was conducted by reviewing the medical records of patients who died in the dermatology ward at a tertiary hospital in Korea in a period of 17 years. Patient demographics, dermatologic diseases, immediate cause of death, comorbidities, and clinical factors related with mortality were investigated.
Results: The total number of inpatients in the dermatology ward during the study period was 740. Thirteen patients (1.76%, 5 men and 8 women) expired while they were admitted in the hospital. The median age of the patients was 63.8 years (range, 18 to 86), and the maximum number of deaths occurred in patients over 70 years old (7 out of 13 patients, 53.8%). Drug reactions were the most common dermatologic complication resulting in admission, followed by bullous disease, generalized pustular psoriasis, and dermatomyositis. Most patients had multiple comorbidities, including hypertension, diabetes mellitus, kidney disease, and angina. Sepsis due to infection was the most common cause of death.
Conclusion: Infection was the most important factor contributing to mortality. Old age, immunosuppression, and comorbidities were important factors that contributed to mortality in dermatologic inpatients.
The study was approved by the Institutional Review Board of (IRB No. 05-2022-094). This study was conducted in accordance with the principles of the Declaration of Helsinki.
Keywords
Death Dermatology Infection Mortality Sepsis
Most dermatologic diseases have very low mortality rates and are generally managed on an outpatient basis. Never- theless, some dermatologic diseases can be life-threatening and require intensive care as a result of the disease itself or its complications. Analysis of dermatologic diseases and their associated risk factors could provide information to predict or prevent patient death. However, only a few studies have assessed the mortality rates among patients with derma- tologic diseases.
We have recorded 13 patient deaths in the dermatology ward from 1998 to 2014. The aim of this study is to in- vestigate dermatologic diseases and the associated factors that lead to death by analyzing these cases.
1. Data collection
This is a retrospective study conducted at a tertiary hospital in Korea (Department of Dermatology at Pusan National University Hospital, Republic of Korea). All data were collected from the medical records of dermatologic inpatients from 1998 to 2014. Patients who died from malignancy, including melanoma and non-melanoma skin cancer, were excluded. This study was approved by the Institutional Review Board of the Pusan National University Yangsan Hospital (IRB No. 05-2022-094).
Patient medical records and death certificate data were retrospectively reviewed. We analyzed the clinical findings according to the following: (1) patient demographic data, including age, sex, and comorbidities; (2) diagnosis of derma- tologic disorders resulting in admission; (3) treatments and progression including length of hospital stay (LoS); (4) cause of death; (5) and laboratory findings, including bacteriological profile and radiologic findings.
2. Statistical analysis
We compared the clinical parameters between expired and recovered patients with Stevens-Johnson syndrome and toxic epidermal necrolysis (SJS/TEN). Statistical analyses were performed using SPSS ver. 21.0 (IBM Co., Armonk, NY, USA). Comparisons of clinical parameters between the groups were performed using Fisher's exact test for categorical variables and Mann-Whitney U test for numerical variables. p-values less than 0.05 were considered statistically significant.
|
No |
Diagnosis |
N (%) |
|
1 |
Herpes zoster |
247 (33.4) |
|
2 |
Drug reaction |
75 (9.9) |
|
3 |
Psoriasis‡ |
36 (4.9) |
|
4 |
Atopic dermatitis |
34 (4.6) |
|
5 |
Contact dermatitis |
31 (4.2) |
|
6 |
Cellulitis/Erysipelas |
31 (4.2) |
|
7 |
Eczema hepeticum |
26 (3.5) |
|
8 |
Pemphigus
vulgaris/foliaceus |
23 (3.1) |
|
9 |
Exfoliative dermatitis |
22 (3.0) |
|
10 |
Varicella |
21 (2.9) |
|
11 |
Erythema multiforme |
20 (2.7) |
|
12 |
Urticaria/Angioedema |
20 (2.7) |
|
13 |
Pyoderma/Abscess/Impetigo |
19 (2.6) |
|
14 |
Vasculitis |
16 (2.2) |
|
15 |
Bullous pemphigoid |
15 (2.0) |
|
16 |
Staphylococcal scalded skin
syndrome |
15 (2.0) |
|
17 |
Eczematous dermatitis |
12 (1.6) |
|
18 |
Hidradenitis suppurativa |
11 (1.5) |
|
19 |
Pyoderma gangrenosum |
9 (1.2) |
|
20 |
Miscellaneous |
57 (7.7) |
|
†Admission for surgical treatment of malignant or benign cutaneous
tumor, laser treatment, and aesthetic treatment was excluded ‡Psoriasis included psoriasis vulgaris, generalized pustular psoriasis, pustulosis palmaris et
plantaris, and acrodermatitis continua of Hallopeau |
||
A total of 740 patients were hospitalized in the derma- tologic ward from 1998 to 2014 (Table 1, admission for the surgical treatment of malignant or benign cutaneous neo- plasia, laser treatment, and aesthetic treatment was excluded). Thirteen patients who eventually died during hospitalization were enrolled in this study. The proportion of expired cases was 1.76% (13 deaths/740 dermatologic inpatients) during the study period. Table 2 shows the patient demographics, comorbidities, and dermatological disorders causing mortality.
|
Patient |
Dermatologic |
Sex |
Age |
Admission- |
Treatment |
Underlying |
Cause of |
Cause of death |
|
|
1 |
Drug |
SJS/TEN |
F |
62 |
11 |
Systemic steroid (PO) |
HTN, |
- |
Myocardial |
|
2 |
SJS/TEN |
F |
18 |
99 |
Systemic steroid Systemic antibiotics |
- |
Pneumonia |
Sepsis |
|
|
3 |
SJS/TEN |
F |
76 |
12 |
Systemic steroid |
HTN, DM, |
Sepsis |
Sepsis |
|
|
4 |
SJS/TEN |
M |
76 |
7 |
Systemic steroid Systemic antibiotics |
DM, |
Sepsis |
Sepsis |
|
|
5 |
SJS/TEN |
M |
58 |
46 |
Systemic steroid Systemic antibiotics |
HTN |
AKI |
Respiratory failure |
|
|
6 |
SJS/TEN |
M |
86 |
33 |
Systemic steroid Systemic antibiotics |
HTN |
Pneumonia |
Respiratory failure |
|
|
7 |
Exanthematous |
F |
73 |
40 |
Systemic steroid |
HTN, DM, |
Pneumonia |
Respiratory failure |
|
|
8 |
Bullous pemphigoid |
F |
56 |
53 |
Systemic steroid Systemic antibiotics |
DM |
Sepsis |
Sepsis |
|
|
9 |
M |
71 |
43 |
Systemic steroid Systemic antibiotics |
DM, HTN, Psoriasis |
Pneumonia |
Respiratory failure |
||
|
10 |
Exfoliative dermatitis |
F |
81 |
29 |
Topical steroid |
HTN |
Pneumonia |
Sepsis |
|
|
11 |
F |
81 |
17 |
Cyclosporine |
HTN, DM |
AKI |
Respiratory failure |
||
|
12 |
Generalized pustular |
F |
37 |
8 |
Methotrexate |
- |
- |
Sepsis |
|
|
13 |
Dermatomyositis (n = 1) |
M |
55 |
31 |
Systemic steroid Hydroxychloroquine |
- |
Increased |
Sepsis |
|
|
AKI: acute kidney injury, CKD: chronic kidney
disease, DM: diabetes mellitus, HTN: hypertension, SJS/TEN: Stevens-Johnson syndrome
and toxic epidermal necrolysis |
|||||||||
1. Demographics
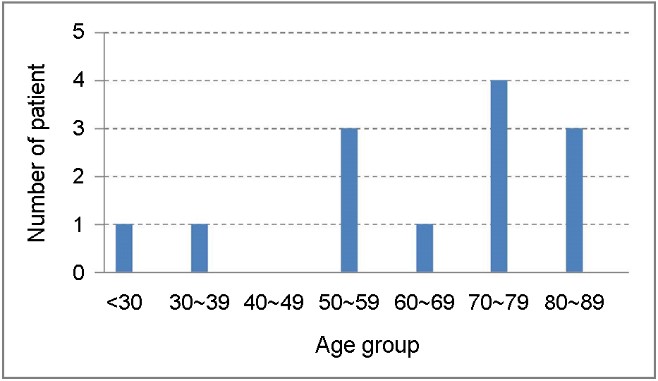
Fig. 1 shows the age distribution of the patients. The mean age was 63.8 years (range, 18~86). Eleven of out 13 patients (84.6%) were older than 55 years, and the highest mortality was recorded in patients older than 70 years (7 out of 13 patients, 53.8%). A slight female dominance among the patients was observed (male : female ratio, 1:1.6). Ten of 13 patients (76.9%) had more than one comorbidity, including hypertension (n = 8), diabetes mellitus (n = 6), angina pectoris (n = 2), and chronic kidney disease (n = 1). None presented with HIV infection.
2. Diagnosis of dermatologic disorders
Dermatologic complications responsible for admissions were drug reactions, including SJS/TEN and exanthematous drug eruption (n = 7), as well as bullous pemphigoid (BP, n = 2), exfoliative dermatitis (n = 2), generalized pustular psoriasis (GPP, n = 1), and dermatomyositis (n = 1).
The offending medications responsible for drug reac- tions were antibiotics in two patients, non-steroidal anti-inflammatory drugs in two patients, allopurinol in two patients, and "unknown" in one patient.
SJS/TEN was the most common dermatologic disorder in this study (n = 6). The mean detached body surface area (BSA) was 42.0 ± 19.2% (range, 20~70), and the mean SCORTEN was 2.8 ± 1.3 (range, 1~4) points upon admission. Compared with the recovered patients with SJS/TEN during the same period (n = 58), expired patients had significantly lower serum albumin levels (expired patients 3.0 ± 0.5 vs. recovered patients 3.9 ± 0.6, Mann-Whitney U test, p = 0.002) and higher blood urea nitrogen levels (expired patients 49.6± 39.7 vs. recovered patients 18.9 ± 9.6, Mann-Whitney U test, p = 0.016). Other laboratory findings, SCORTEN, and BSA were not significantly different between the two groups (data not shown).
3. Treatments and progression
Twelve patients (92.3%) were treated with immunosup- pressive agents, such as systemic steroids (n = 10, 76.9%), cyclosporine (n = 1, 7.7%), and methotrexate (n = 1, 7.7%). Antibiotics were administered in seven patients (53.8%) from admission. None of the expired patients with SJS/TEN were treated with intravenous immunoglobulin.
Two patients (15.4%) died in the dermatology ward, and 11 patients (84.6%) were transferred to another department or an intensive care unit. A total of 11 patients were trans- ferred because of suspected sepsis (n = 6), continuous fever (n = 2), renal dysfunction (n = 2), or abnormal liver function (n = 1). The average LoS in hospital (from admission to expiration) of all patients was 31.6 ± 24.7 days (range, 7~99). The mean LoS in dermatology wards (from admission to expiration or transfer) was 9.5 ± 8.5 days.
4. Cause of death
Seven of 13 patients (53.8%) developed septic shock and died as a result. Five patients (38.5%) died from acute respiratory failure caused by pneumonia (n = 4) or diffuse alveolar hemorrhage (n = 1). One patient (7.7%) died from acute myocardial infarction.
5. Laboratory findings
Abnormal laboratory findings upon admission included leukocytosis (n = 5), electrolyte imbalance (n = 3), throm- bocytopenia (n = 2), anemia (n = 2), abnormal kidney function (n = 2), and abnormal liver function (n = 2).
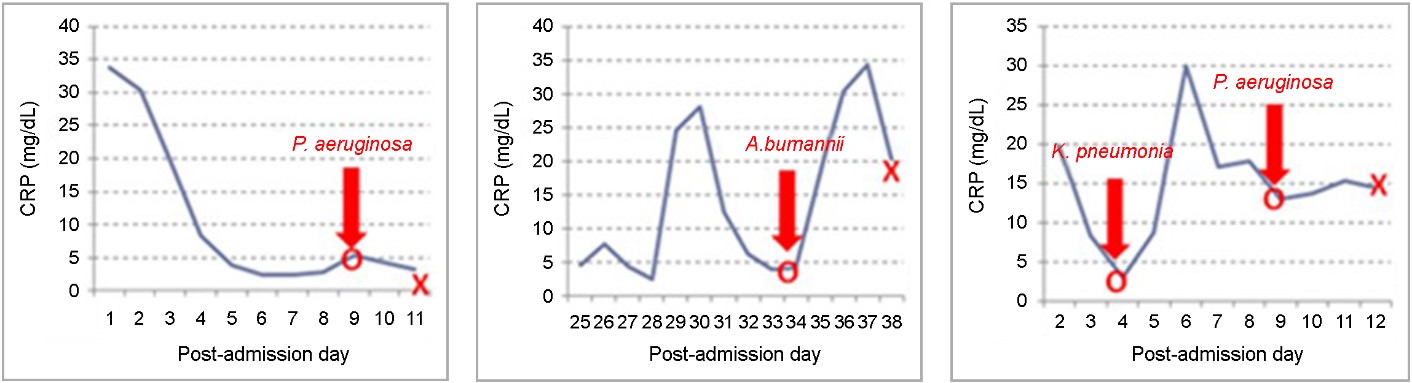
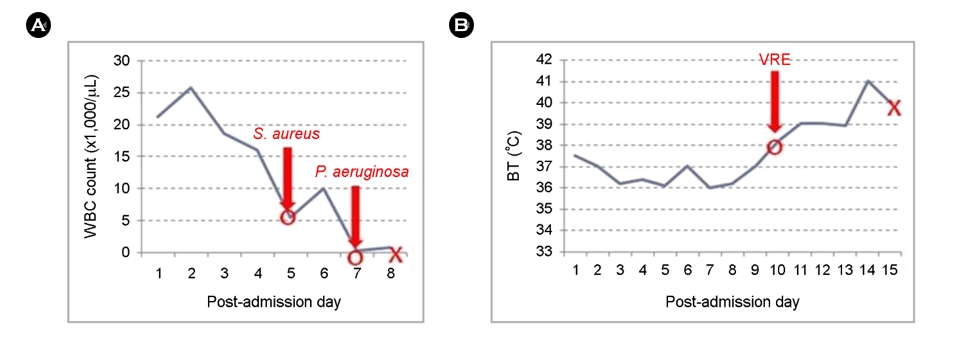
Infection developed in 11 patients (84.6%). The most com- mon infection site was the respiratory tract (pneumonia), followed by the kidneys (acute pyelonephritis) and skin (soft tissue infection). Table 3 summarizes the results of the blood cultures. Twelve microorganisms in 7 patients were identified using bacterial cultures from blood. The most common patho- gens were well-known microorganisms causing hospital-acquired infections (HAI), such as Pseudomonas aeruginosa, Staphylococcus aureus, Klebsiella pneumoniae, and Acineto- bacter baumannii. In most cases, the results of the blood cultures were reported after the patient's death. Among the patients who died from infection, a reversal of C-reactive proteins (CRP) from decreasing to increasing levels with positive microbial growth in the blood cultures was observed in three patients (Fig. 2). Sudden-onset neutropenia (Fig. 3A) and sudden-onset fever (Fig. 3B) with positive microbial growth in the blood cultures were observed in one patient.
|
Pathogen |
Number |
|
Pseudomonas aeruginosa |
6 |
|
Staphylococcus aureus (including methicillin-resistant |
2 |
|
Klebsiella pneumonia |
1 |
|
Acinetobacter baumannii |
1 |
|
Vancomycin-resistant |
1 |
|
Candida albicans |
1 |
The frequency of dermatologic diseases causing death is different for each country. The most common diseases were autoimmune blistering diseases in Brazil1 and India2, chronic ulcers and cellulitis in the United States3, and SJS/TEN in the United Kingdom4. In the present study, SJS/TEN was the most common disorder resulting in death, followed by BP, exfoliative dermatitis, GPP, and dermatomyositis in derma- tologic inpatients.
SJS/TEN is a well-known, life-threatening disorder. The overall hospital mortality rate of SJS/TEN is 22~25%5. In our study, the mortality rate of SJS/TEN was 9.4% (6 out of 64 patients with SJS/TEN). The prognosis score of SJS/TEN (SCORTEN) is useful, but the SCORTEN upon admission was not significantly different between the expired and recovered patients (expired patients 2.8 vs. recovered patients 2.0, Mann-Whitney U test, p = 0.224) in the present study. BP is the most common autoimmune blistering disease. Old age, poor general health, and the presence of anti-BP180 anti- bodies have been associated with a poor prognosis5. Exfoliative dermatitis is potentially life-threatening because of compli- cations, including fluid and electrolyte imbalance, thermo- regulatory disturbance, fever, tachycardia, high-output failure, and hypoalbuminemia. GPP can potentially result in life-threatening complications, such as hypocalcemia, bacterial superinfection, sepsis, and dehydration. Death occurred in 2% of the GPP cases6. The overall mortality rate of dermato- myositis and polymyositis was 22%. Variables associated with poor outcomes of dermatomyositis and polymyositis were older age, pulmonary and esophageal involvement, and cancer7.
It is well-known that old age contributes to increased mortality in diseases. In a population-based study examining death from non-neoplastic skin disease in the United States, most deaths occurred in patients aged 65 years and older3. Similarly, in the present study, 85% of deaths occurred in patients aged 55 years and older, and the highest mortality was reported in patients aged 70~79.
Infection developed in 11 patients (84.6%) and 7 patients (53.8%) died from septic shock. Inflammation, fissuring, and excoriation of the diseased skin increase susceptibility to bacterial colonization and infection in dermatologic diseases, such as SJS/TEN, exfoliative dermatitis, and blistering diseases5. Immunosuppressive agents are the mainstay of treatment in severe inflammatory skin diseases, but they can also predis- pose patients to infections8. Most patients (92.3%) were treated with immunosuppressive agents in the present study.
The most common pathogens were P. aeruginosa, S. aureus (including MRSA), K. pneumoniae, and E. coli, which are important microorganisms in HAI. Tao et al.9 reported that P. aeruginosa, K. pneumoniae, and E. coli are common pathogens in HAI. Vijayamohan et al.10 reported a high pre- valence of methicillin-resistant S. aureus (MRSA) infection among dermatology inpatients. Prolonged duration of illness, prolonged antibiotic and steroid therapy, and diabetes were relative risk factors for acquiring MRSA. In another study, prolonged LoS was an important factor in hospital-acquired MRSA11. Therefore, precautions against HAI during hospital- ization, such as hand decontamination, personal hygiene, disinfection of patient equipment, and safe injection prac- tices12, could decrease mortality among patients with severe dermatologic disease.
Among the patients who died from sepsis, sudden-onset fever, neutropenia, and CRP elevation were observed. Bor et al.13 reported a fourfold increase in the mortality rate of patients with fever compared with those without fever. CRP is an acute-phase reactant, suggesting that its levels rise in response to inflammation. In sepsis, IL-6, TNF-a, and IL-1b stimulate the release of CRP during the inflammatory cascade14,15. Therefore, the reversal of CRP from decreasing to increasing levels could be a marker of infection, and serial CRP measurement could be a valuable tool in patients with severe dermatologic disorders. Leukopenia (defined as a white blood cell count of <4,000/μL) can be seen in sepsis as one of the criteria of systemic inflammatory response syndrome. Leukopenia indicates anomalies in the host's inflammatory response and is associated with poor outcomes16. Therefore, although fever, CRP elevation, and neutropenia can be observed without infection, such as those with connective tissue disease, malignancy, and cardiovascular disease17,18, efforts should be made to identify any infection sites in these situations.
The limitations of this study include its retrospective design, small cohort size, and the single center nature of the study.
In conclusion, our study revealed that drug reactions, such as SJS/TEN, were the most common dermatologic disease leading to mortality that is associated with infection and sepsis. Factors including old age, epithelial loss by derma- tologic diseases, and immunosuppression could predispose infection in these patients. Infection, especially during hos- pitalization, was the most important factor contributing to mortality. Therefore, efforts to prevent HAI could contribute to decreasing mortality. This study aims to improve the under- standing of severe dermatologic diseases and contributes to the prevention of deaths in patients with dermatologic disorders.
References
1. de Paula Samorano-Lima L, Quiterio LM, Sanches JA Jr, Neto CF. Inpatient dermatology: profile of patients and characteristics of admissions to a tertiary dermatology inpatient unit in Sao Paulo, Brazil. Int J Dermatol 2014; 53:685-691
Google Scholar
2. Nair PS, Moorthy PK, Yogiragan K. A study of mortality in dermatology. Indian J Dermatol Venereol and Leprol 2005;71:23-25
3. Lott JP, Gross CP. Mortality from nonneoplastic skin dis- ease in the United States. J Am Acad Dermatol 2014; 70:47-54
Google Scholar
4. George SM, Harrison DA, Welch CA, Nolan KM, Friedmann PS. Dermatological conditions in intensive care: a secondary analysis of the Intensive Care National Audit and Research Centre (ICNARC) Case Mix Pro- gramme database. Crit Care 2008;12(Suppl 1):S1
Google Scholar
5. Mockenhaupt M, Roujeau J. Epidermal necrolysis (Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis). In: Kang S, Amagai M, Bruckner AL, Enk AH, Margolis DJ, McMichael AJ, editors. Fitzpatrick's Dermatology. 9th ed. New York: McGraw-Hill, 2019:733-748
Google Scholar
6. Augey F, Renaudier P, Nicolas JF. Generalized pustular psoriasis (Zumbusch): a French epidemiological survey. Eur J Dermaltol 2006;16:669-673
Google Scholar
7. Marie I, Hachulla E, Hatron PY, Hellot MF, Levesque H, Devulder B, et al. Polymyositis and dermatomyositis: short term and longterm outcome, and predictive factors of prognosis. J Rheumatol 2001;28:2230-2237
Google Scholar
8. Ozer B, Ozbakis Akkurt BC, Duran N, Onlen Y, Savas L, Turhanoglu S. Evaluation of nosocomial infections and risk factors in critically ill patients. Med Sci Monit 2011; 17:PH17-22
Google Scholar
9. Tao XB, Qian LH, Li Y, Wu Q, Ruan JJ, Cai DZ, et al. Hospital-acquired infection rate in a tertiary care teaching hospital in China: a cross-sectional survey involving 2434 inpatients. Int J Infect Dis 2014;27:7-9
Google Scholar
10. Vijayamohan N, Nair SP. A study of the prevalence of methicillin-resistant Staphylococcus aureus in dermatology inpatients. Indian Dermatol Online J 2014;5:441-445
11. Wong JG, Chen MI, Win MK, Ng PY, Chow A. Length of stay an important mediator of hospital-acquired methicillin-resistant Staphylococcus aureus. Epidemiol Infect 2016;144:1248-1256
Google Scholar
12. Mehta Y, Gupta A, Todi S, Myatra S, Samaddar DP, Patil V, et al. Guidelines for prevention of hospital acquired infections. Indian J Crit Care Med 2014;18:149-163
Google Scholar
13. Bor DH, Makadon HJ, Friedland G, Dasse P, Komaroff AL, Aronson MD. Fever in hospitalized medical patients: characteristics and significance. J Gen Intern Med 1988; 3:119-125
Google Scholar
14. Kushner I, Jiang SL, Zhang D, Lozanski G, Samols D. Do post-transcriptional mechanisms participate in induction of C-reactive protein and serum amyloid A by IL-6 and IL-1? Ann N Y Acad Sci 1995;762:102-107
Google Scholar
15. Gabay C, Kushner I. Acute-phase proteins and other systemic responses to inflammation. N Engl J Med 1999; 340:448-454
Google Scholar
16. Kreger BE, Craven DE, McCabe WR. Gram-negative bacteremia. IV. Re-evaluation of clinical features and treatment in 612 patients. Am J Med 1980;68:344-355
Google Scholar
17. Göktay F, Ceran N, Aydingöz IE, Mansur AT. Characteristics of fever, etiologic factors, antibiotic use and prognosis in febrile dermatology inpatients. Int J Dermatol 2013; 52:1331-1337
Google Scholar
18. Limper M, de Kruif MD, Duits AJ, Brandjes DP, van Gorp EC. The diagnostic role of procalcitonin and other bio- markers in discriminating infectious from non-infectious fever. J Infect 2010;60:409-416
Google Scholar
Congratulatory MessageClick here!