pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed

pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed
Taekwoon Kim,Yongwoo Choi,Jong Soo Choi,Joonsoo Park
10.17966/JMI.2022.27.1.14 Epub 2022 March 31
Abstract
Most superficial fungal infections of the skin are caused due to dermatophytosis or yeasts. However, it is rare for Cryptococcus species to cause infections. Among the fungal infections by Cryptotoccus species, Cryptococcus (C.) neoformans and C. gattii are the most common pathogenic species. However, other rare infections have been reported. Subcutaneous cryptococcosis due to Naganishia (N.) diffluens (formerly Cryptococcus diffluens) was first reported in Turkey. We present the case of a 53-year-old man who had mild pruritic annular, erythematous, scaly patches with inner small, crusted papules and nodules on both axilla and trunk for 3 months. A potassium hydroxide test on his abdomen scales was positive. Fungal culture, light microscopic findings, and the sequencing of an internal transcribed spacer of rRNA gene confirmed the presence of N. diffluens. As a result we diagnosed the patient with a superficial fungal infection caused by N. diffluens and treated it successfully with oral itraconazole and topical isoconazole.
PATIENT CONSENT STATEMENT:The patient provided written informed consent for the publication and the use of his images.
Keywords
Superficial fungal infections are some of the most common dermatologic diseases seen worldwide. It is estimated that 13.8~20% of the population has had a dermatophyte infection, and approximately 12~13% has onychomycosis1. Trichophyton (T.) rubrum, T. tonsurans, T. mentagrophytes, Microsporum (M.) canis are the most common causative agents of superficial fungal infection in the US1.
Other species, such as Cryptococcus species, can also cause superficial fungal infection1.
Cryptococcus species are basidiomycetous yeasts that are thought to be the cause of a variety of diseases2-4. Crypto- coccus (C.) neoformans and C. gattii are the most common pathogenic species2-4. However, the incidence of infection due to non-neoformans cryptococcal species such as Naganishia (N.) albida and Papiliotrema laurentii (formerly C. laurentii) has increased recently5,6. Other very rare infections including N. diffluens (formerly C. diffluens) have also been reported (Table 1)7 . We present a case of N. diffluen-sinduced superficial fungal infection.
|
No |
Age/sex/year |
Location |
Host status |
Examination |
Treatment |
Outcome |
|
1 |
17/Male/2007 |
Turkey |
Not indicated |
Culture, biopsy, |
Itraconazole |
Cured |
|
2 |
53/Male/2021 |
Korea |
Alcoholic liver cirrhosis, Hypertension |
Culture, DNA sequencing |
Itraconazole |
Cured |
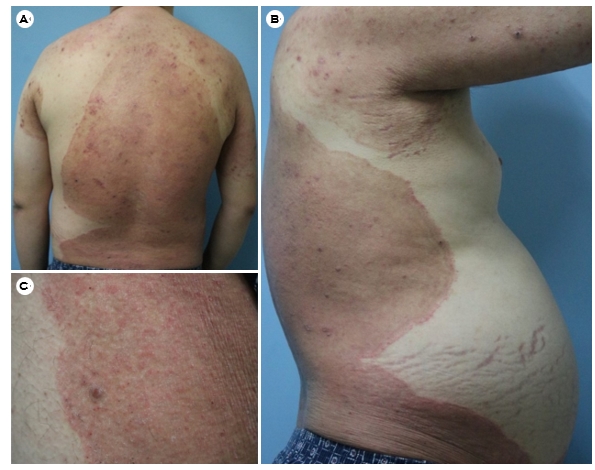

A 53-year-old man presented to our clinic with a pruritic skin rash on his trunk and axilla.
For three months, he had mildly pruritic annular, erythematous, scaly patches with inner small, crusted papules and nodules on both axilla and trunk (Fig. 1). The lesions enlarged after the application of topical corticosteroids, administered by a local medical center approximately 2 months ago. Additionally, he suffered from hypertension and alcoholic liver cirrhosis. The patient had recurrent ascites and a history of being hospitalized in a bed-ridden state regularly. Furthermore, because of his low socio-economic status, his surroundings were unclean and humid.
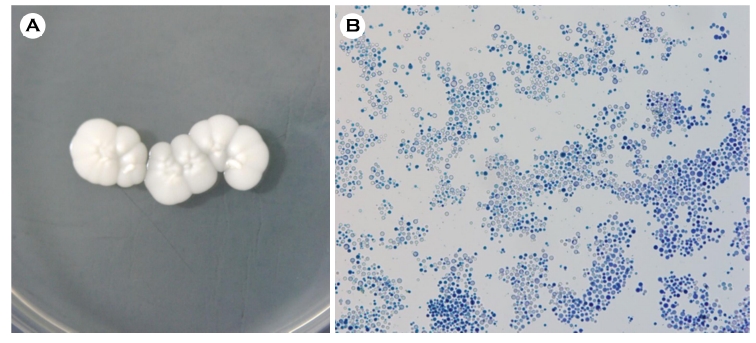
A potassium hydroxide test performed on the scales on the lesions of multiple sites was all positive, and multiple round yeast cells were observed. Specimens were collected at least twice from multiple lesions and cultured at 28℃ for two weeks on Sabouraud dextrose agar. All of the agars produced white and creamy colonies with smooth surfaces (Fig. 2A). Lactophenol cotton blue staining of the smears showed yeast cells similar to Cryptococcus under the micro- scope (Fig. 2B). The presence of N. diffluens was finally confirmed through sequencing of the rRNA gene' internal transcribed spacer (ITS) (Fig. 3). Furthermore, no evidence of systemic infection was discovered in X-ray, CT, or laboratory findings. Thus, we diagnosed the patient with a superficial fungal infection caused by N. diffluens. For two weeks, the patient was treated with an oral antifungal agent (itracona- zole, 100 mg) and a topical antifungal agent (isoconazole), and the lesions significantly improved. There was no evidence of recurrence for 6 months.
Most rare Cryptococcus species belong to the C. albidus clade (N. adeliensis, N. diffluens, N. liquefaciens, Filobasidium magnus, and N. uzbekitanensis). They were misidentified as N. albida before molecular identification (formerly C. albidus). The ITS region and the 28S D1/D2 region of rRNA DNA sequence analysis reclassified them as N. diffluens8.
The genus Naganishia has asexually reproducing yeast species. Naganishia species are basidiomycetous yeasts and are characterized by the lack of basidiocarps. Typically, hyphae and pseudohyphae are absent, while budding cells and ballistoconidia are present9. N. diffluens are globose or elliptical, encapsulated yeast cells of varying sizes8. The histopathological features of N. diffluens are similar to those of cryptococcal infection, and a phenol oxidase test that is negative can aid in the differentiation from C. neoformans, the most common cause of cryptococcosis. Biopsy, culture, and molecular analysis are required for a definitive diagnosis. For accurate identifi- cation of N. diffluens isolates, DNA sequence analysis of the ITS region and the 28S D1/D2 region of rRNA are necessary7.
Typically, primary cutaneous infections caused by Crypto- coccus species occur infrequently as a result of organisms being directly inoculated into the skin by a parasitized object. The disease's clinical manifestation is determined by the host's response, the size of the inoculum, and the organism's innate virulence. Cryptococcosis can present with a variety of skin and soft tissue manifestations including purpura, vesicles, nodules, abscesses, ulcers, acneiform eruptions, granulomas, pustules, draining sinuses, and cellulitis10,11. The majority of skin and soft tissue manifestations occur in the context of a disseminated disease, which is seen in 10~15% of patients with systemic cryptococcosis11. The symptoms of systemic cryptococcosis can range from a mild fever and cough to meningitis, osteomyelitis, pyelonephritis, prostatitis, hepatitis, and peritonitis10. Thus far, most of the reported cases have been by C. neoformans. Subcutaneous cryptococcosis due to N. diffluens (formerly C. diffluens) was first reported in Turkey (Table 1)7. In that case, the patient has a two-month history of nontender nodules that began on the right wrist and progressed to the right arm. The patient was treated through oral itraconazole for 3 months after diagnosis through culture, biopsy, and DNA sequence analysis. In addition to N. diffluens, the first case of N. albida superficial infection was reported in Iran12. In that case, the patient has been suffering from a hyperpigmented patch on his axilla for more than a month. The patient was treated through oral itraconazole for 10 days after diagnosis through culture and DNA sequence analysis. The case described here is the first report of a superficial infection caused by N. diffluens in South Korea. The patient's alcoholic liver cirrhosis and long-term bed-ridden condition as a result of cirrhosis compli- cations are thought to be risk factors for the superficial fungal infection.
The first choice of treatment for cryptococcosis is the combination of amphotericin B and flucytosine13,14. However, the treatment of N. diffluens superficial infection is not well defined. Oral itraconazole was used successfully in reported cases, including this one7, and in vitro susceptibilities of N. diffluens to several antifungal agents (amphotericin B, flu- cytosine, fluconazole, itraconazole, voriconazole, and posa- conazole) revealed that voriconazole and posaconazole have good activity against N. diffluens15.
Unfortunately, the biopsy was not performed in this case. However, it is of diagnostic significance in that the test (including KOH, culture, molecular analysis) conducted on the multiple sites of lesion produced the same results. The ability of N. diffluens to cause superficial infection has been discovered through this report and literature reviews. As a result, we present a rare case of N. diffluens caused superficial fungal infection.
References
1. Long SS, Pickering LK, Prober CG. Principles and practice of pediatric infectious diseases. 4th ed. Philadelphia: Saunders, 2012:1246-1250
Google Scholar
2. Faress JA, Hejal R. Pulmonary crytpococcosis due to C. albidus in a patient on Infliximab. Chest 2004;126:987s-988s
Google Scholar
3. de Castro LE, Sarraf OA, Lally JM, Sandoval HP, Solomon KD, Vroman DT. Cryptococcus albidus keratitis after corneal transplantation. Cornea 2005;24:882-883
Google Scholar
4. Hagen F, Lumbsch HT, Arsic Arsenijevic V, Badali H, Bertout S, Billmyre RB, et al. Importance of resolving fungal nomenclature: the case of multiple pathogenic species in the Cryptococcus genus. mSphere 2017;2: e00238-17
5. Burnik C, Altintas ND, Ozkaya G, Serter T, Selçuk ZT, Firat P, et al. Acute respiratory distress syndrome due to Cryptococcus albidus pneumonia: case report and review of the literature. Med Mycol 2007;45:469-473
Google Scholar
6. Khawcharoenporn T, Apisarnthanarak A, Mundy LM. Non-neoformans cryptococcal infections: a systematic review. Infection 2007;35:51-58
Google Scholar
7. Kantarcioğlu AS, Boekhout T, de Hoog GS, Theelen B, Yücel A, Ekmekci TR, et al. Subcutaneous cryptococcosis due to Cryptococcus diffluens in a patient with sporo- trichoid lesions case report, features of the case isolate and in vitro antifungal susceptibilities. Med Mycol 2007; 45:173-181
Google Scholar
8. Morales-López SE, Garcia-Effron G. Infections due to rare Cryptococcus species. A literature review. J Fungi (Basel) 2021;7:279
Google Scholar
9. Francisco EC, de Jong AW, Hagen F, Cryptococcosis and Cryptococcus. Mycopathologia 2021;186:729-731
Google Scholar
10. Dimino-Emme L, Gurevitch AW. Cutaneous manifestations of disseminated cryptococcosis. J Am Acad Dermatol 1995;32:844-850
Google Scholar
11. Anderson DJ, Schmidt C, Goodman J, Pomeroy C. Cryp-tococcal disease presenting as cellulitis. Clin Infect Dis 1992;14:666-672
Google Scholar
12. Aghaei Gharehbolagh S, Nasimi M, Agha Kuchak Afshari S, Ghasemi Z, Rezaie S. First case of superficial infection due to Naganishia albida (formerly Cryptococcus albidus) in Iran: A review of the literature. Current Med Mycol 2017;3:33-37
Google Scholar
13. Huang W, Liao G, Baker GM, Wang Y, Lau R, Paderu P, et al. Lipid flippase subunit Cdc50 mediates drug resistance and virulence in Cryptococcus neoformans. mBio 2016; 7:e00478-16
Google Scholar
14. Smith KD, Achan B, Hullsiek KH, McDonald TR, Okagaki LH, Alhadab AA, et al. Increased antifungal drug re- sistance in clinical isolates of Cryptococcus neoformans in Uganda. Antimicrob Agents Chemother 2015;59:7197- 7204
Google Scholar
15. Kantarcioglu AS, Boekhout T, Yücel A, Altas K. Suscep- tibility testing of Cryptococcus diffluens against ampho- tericin B, flucytosine, fluconazole, itraconazole, vori- conazole and posaconazole. Med Mycol 2009;47:169-176
Google Scholar
Congratulatory MessageClick here!