pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed

pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed
Md Shamsuzzaman,Jungmin Kim
10.17966/JMI.2026.31.1.8 Epub 2026 March 31
Abstract
Skin dysbiosis, characterized by microbial imbalance and pathogen overgrowth, plays a central role in several common skin disorders, including acne vulgaris, atopic dermatitis, and chronic wound infections. Conventional broad-spectrum antibiotics often disrupt beneficial skin microbiota and contribute to antimicrobial resistance, including emergence of multidrug-resistant pathogens. These limitations have driven interest in precision antimicrobial strategies. Bacteriophages and phage-derived endolysins have emerged as promising alternatives due to their high specificity, potent bactericidal activity, and ability to disrupt biofilms while preserving commensal bacteria. This review outlines the mechanistic basis and therapeutic potential of phage and endolysin-based approaches for skin diseases. Lytic phages selectively infect and kill bacterial hosts through intracellular replication and cell lysis. In contrast, endolysins enzymatically degrade peptidoglycan, leading to rapid bacterial destruction. The study discusses disease-specific applications, including targeting pro-inflammatory Cutibacterium acnes phylotypes in acne vulgaris, selective reduction of Staphylococcus aureus colonization in atopic dermatitis, and disruption of polymicrobial biofilms involving methicillin-resistant S. aureus and Pseudomonas aeruginosa in chronic wounds. Preclinical studies, early human studies, and case reports suggest reduced pathogen burden and clinical improvement, supporting further clinical evaluation. Remaining challenges include formulation stability, host immune interactions, regulatory pathways, and biomarker-guided clinical trial design. Overall, phage and endolysin-based therapies represent precision, next-generation tools for restoring skin microbial balance in the era of antibiotic resistance.
Keywords
Acne vulgaris Atopic dermatitis Bacteriophages Biofilms Endolysins Skin microbiome
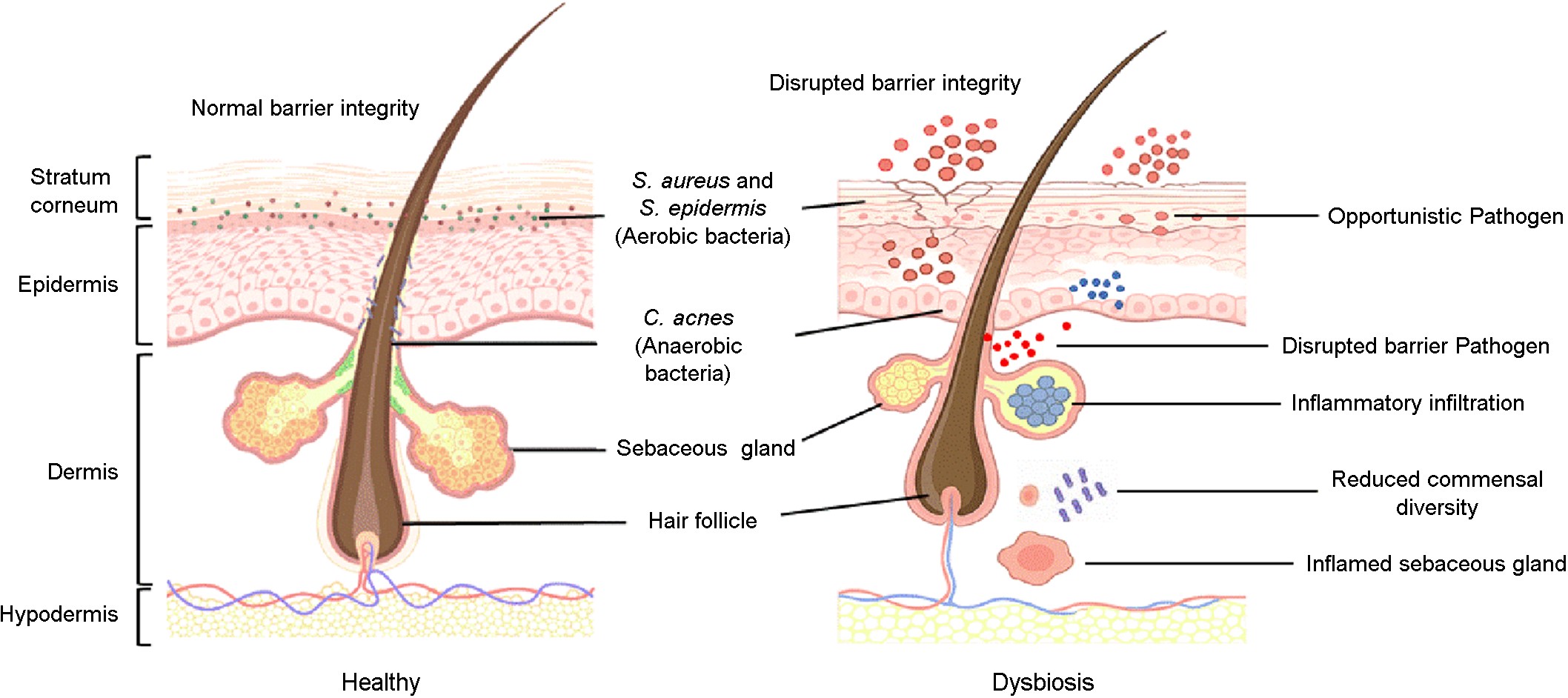
Human skin is a complex ecological interface in which host immunity interacts with a diverse microbial community to maintain barrier integrity and protect against pathogens. Disruption of this balance by inflammation, environmental stress, or antibiotic exposure can lead to microbial dysbiosis, which underlies several common dermatologic conditions, including acne vulgaris, atopic dermatitis (AD), and chronic wound infections1. Overgrowth of pathogenic bacteria, such as C. acnes, S. aureus, methicillin-resistant S. aureus, and P. aeruginosa, amplifies inflammation, impairs barrier function, and contributes to development of recurrent or treatment-resistant disease2,3.
Systemic and topical antibiotics have long been central to management of skin infections and inflammatory dermatoses. However, increasing antimicrobial resistance, disruption of commensal microbiota, and limited efficacy against biofilm-associated bacteria have highlighted the need for more selective antimicrobial strategies4. Phages and phage-derived endolysins have therefore emerged as promising alternatives because they exhibit host-specific bacteriolytic activity, disrupt biofilms, and largely spare beneficial microbiota, making them particularly attractive for dermatologic applications5. Preclinical and early translational studies show that C. acnes-specific phages reduce inflammatory lesions in acne models. Staphylo- coccal endolysins improve outcomes associated with AD while preserving commensal Staphylococcus species. In chronic wounds, phages targeting P. aeruginosa effectively disrupt and degrade bacterial biofilms6-8.
Advances in metagenomic sequencing have further revealed that skin virome, particularly phages, play a significant role in shaping microbial community structure. Phage populations' alterations observed in acne and AD suggest that rather than merely reflecting microbial imbalance, phage bacteria interactions may contribute directly to disease phenotypes9. These findings support the concept of phage-based thera- peutics as precision tools capable of modulating dysbiotic microbial networks rather than indiscriminately eliminating skin bacteria.
This review integrates mechanistic insights, disease-specific evidence, and current therapeutic progress in phage and endolysin-based interventions for acne, AD, and chronic wound infections. It also highlights key opportunities and challenges for translating these biologics into routine dermato- logic practice.
The skin microbiome is a highly organized ecosystem of bac- teria, fungi, viruses, and phages occupying distinct anatomical niches. Sebaceous regions are dominated by Cutibacterium species, moist areas are enriched with Staphylococcus and Corynebacterium, and dry skin sites support greater microbial diversity10. This spatial distribution reflects metabolic speciali- zation and continuous interactions between resident microbes and a host immune system. Under homeostatic conditions, commensal microorganisms contribute to barrier integrity, induce antimicrobial peptide production, and support immune tolerance11.
Dysbiosis refers to disruptions in microbial composition, functional activity, or phage bacteria interactions that dis- turb this balance and promote disease. In acne vulgaris, inflammatory C. acnes phylotypes, particularly IA1 and IA2, become dominant within the pilosebaceous unit. These strains produce porphyrins, extracellular lipases, and biofilms that enhance oxidative stress and follicular inflammation12. Multi- locus sequence typing analyses of Korean clinical isolates further demonstrate marked strain-level diversity and emerging antibiotic resistance in C. acnes, emphasizing limitations of prolonged antibiotic therapy and the need for targeted alter- natives13.
AD displays a distinct dysbiotic pattern. Disease flares are associated with excessive colonization by S. aureus and a concurrent loss of protective commensal staphylococci. This imbalance promotes Th2-skewed immune responses, epi- dermal barrier dysfunction, and increased susceptibility to secondary infections. Recent metagenomic studies have also revealed pronounced alterations in AD patients' skin virome3,14. Changes in phage abundance and composition suggest that rather than merely reflecting inflammatory states, phage-bacterium interactions may actively influence microbial domin- ance and disease severity15.
Chronic wounds present another form of severe dysbiosis. Stable polymicrobial biofilms containing MRSA, P. aeruginosa, enterococci, and anaerobic bacteria characterize such lesions16. Biofilm-associated extracellular matrices limit antibiotic penetration, impair immune clearance, and sustain chronic inflam- mation, ultimately delaying tissue repair.
Together, these findings establish dysbiosis as a central driver of skin disease. Defining microbial and viral community shifts across acne, AD, and chronic wounds provides a critical foundation for precision therapeutics. In this context, phages and endolysins offer targeted strategies to eliminate pathogens while preserving the skin's broader microbial ecosystem17. Fig. 1 illustrates commensal microbes' spatial organization in healthy skin and characteristic shifts observed during dysbiosis across major skin diseases.
Phages exert therapeutic effects through precise bacterial targeting, efficient lytic activity, and minimal disruption of commensal microbiota. The phage life cycle begins with re- cognition and binding to specific bacterial surface receptors that determine host specificity. In skin-associated pathogens, these receptors include lipoteichoic acids on S. aureus, cell wall–associated surface structures on C. acnes, and type IV pili on P. aeruginosa, which serve as essential adsorption receptors for phage PA1Ø6. After attachment, phages inject their genetic material into the host cell, thus leading to take- over of bacterial metabolism, intracellular replication, and eventual host lysis. Newly released virions then infect neigh- boring bacteria, allowing localized amplification at the infection site18,19. Fig. 2 summarizes the lytic phage life cycle's key steps and phage-derived endolysins' bacteriolytic action against skin-associated pathogens.
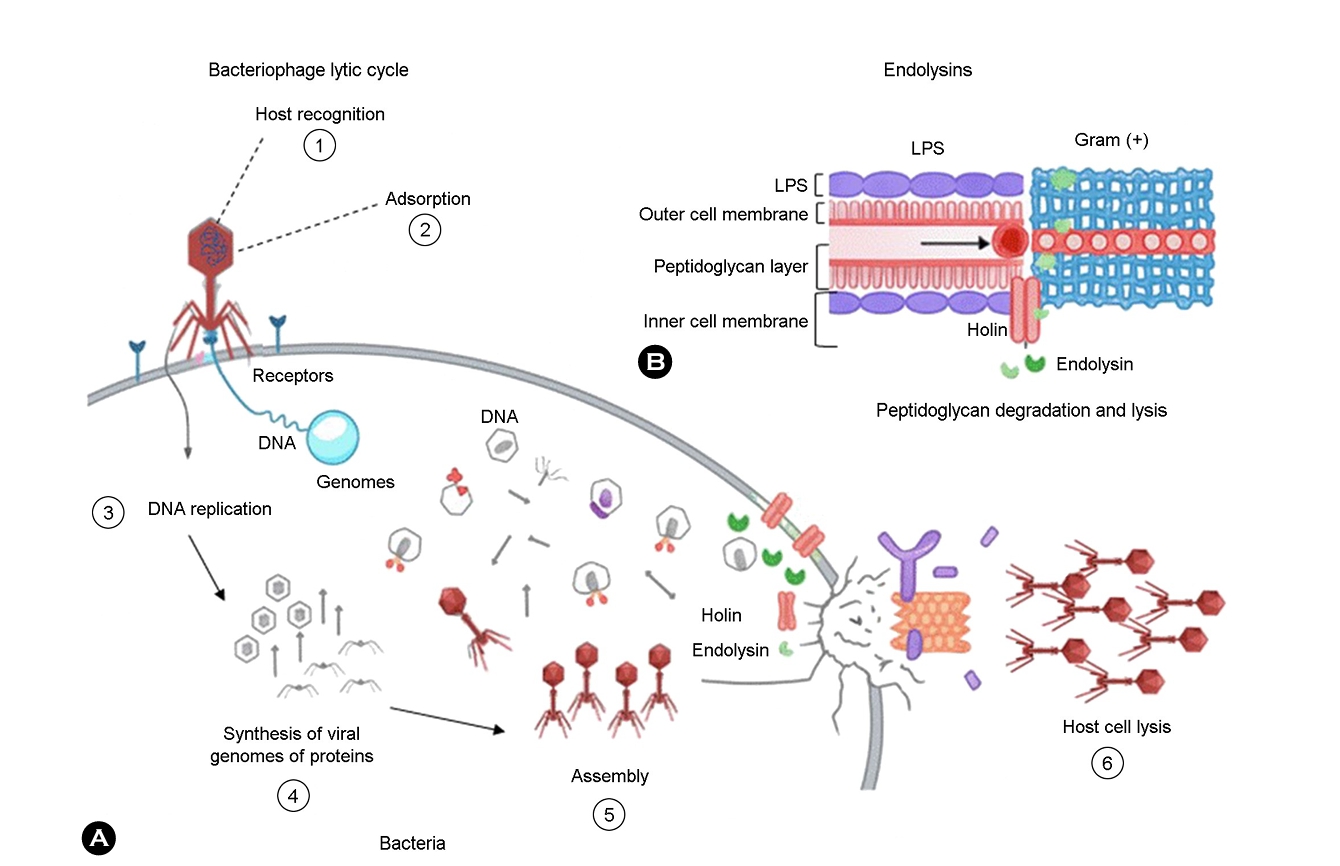
Endolysins represent a complementary antimicrobial strategy. These phage-derived enzymes directly cleave peptidoglycan, leading to rapid bacterial lysis. Their activity is particularly effective against Gram-positive organisms and does not re- quire specific surface receptors or active bacterial growth. This allows endolysins to bypass several biofilm-associated resistance mechanisms. CHAP-domain endolysins display strong lytic activity against S. aureus and related pathogens. They remain stable across broad pH and temperature ranges and retain function in complex biological environments, making them suitable for topical dermatologic use20,21. In in-vitro MRSA biofilm assays, phage SAP-26 shows potent antibiofilm activity and synergizes with rifampicin, whereas its derived endolysin LysSAP26 displays strong bacteriolytic activity, supporting potential translational relevance of SAP-26–based phage and endolysin strategies for chronic wound infection models22,23.
Beyond killing bacteria directly, phages and endolysins offer important ecological advantages. Their narrow host range enables preservation of beneficial commensals, such as S. epidermidis, contributing to immune regulation and barrier repair. Phages can also penetrate mature biofilms through enzymatic degradation of extracellular matrices or replication within deeper biofilm layers, overcoming conventional anti- biotics' significant limitation24. In acne models, lytic C. acnes phages demonstrate strain-selective killing and reduce inflam- matory cytokines' production, suggesting both antimicrobial and immunomodulatory effects25.
Collectively, these mechanisms highlight phage and endolysin-based therapies' suitability for skin diseases. Their precision, antibiofilm activity, and microbiome compatibility position them as next-generation biologics for correcting dys- biosis while minimizing collateral damage17. Table 1 provides a comparative overview of disease-specific targets, therapeutic agents, mechanisms of action, and key outcomes for phage- and endolysin-based interventions across major skin diseases.
|
Skin |
Target |
Therapeutic |
Mechanism of action |
Preclinical and |
References |
|
Acne |
Cutibacterium
acnes
(inflammatory phylotypes) |
C. acnes-specific lytic phages and phage-derived
endolysins |
Phages selectively infect and lyse |
Reduced C. acnes burden, disruption of biofilms, and attenuation of proinflammatory
cytokine responses. |
7, 27 |
|
Atopic |
Staphylococcus
aureus |
S. aureus-specific |
Endolysins selectively cleave |
Decreased S. aureus |
8, 34 |
|
Chronic |
MRSA, Pseudomonas
aeruginosa (polymicrobial biofilms) |
Lytic phages (e.g., |
Phages lyse biofilm-embedded |
Demonstrated reduction of bacterial burden and |
6, 22, 23, |
1. Bacteriophage and endolysin therapy for acne vulgaris
Acne vulgaris arises from complex interplay of follicular hyperkeratinization, excess sebum production, microbial dys- biosis, and cutaneous inflammation. A key microbial feature of acne is the shift in C. acnes populations from a diverse community toward dominance of inflammatory phylotypes, particularly IA1 and IA2. These strains produce porphyrins, extracellular lipases, and biofilms that intensify oxidative stress, promote inflammation, and disrupt follicular barrier function7,12. Multilocus sequence typing studies of Korean clinical isolates further reveal pronounced strain-level hetero- geneity and increasing resistance to commonly used anti- biotics. These findings highlight long-term tetracycline and macrolide therapy's limitations and underscore the need for targeted alternatives13,26.
C. acnes specific phages have emerged as promising pre- cision therapeutics for acne treatment because they exhibit strong lytic activity against acne-associated strains and display highly conserved genomes, suggesting favorable safety pro- files and low risk of horizontal gene transfer27,28. In preclinical acne models, lytic phages' administration reduces bacterial burden and attenuates inflammatory responses. Murine studies demonstrate decreased neutrophil infiltration and reduced inflammatory lesion size following intralesional phage treatment, supporting antimicrobial activity and suggesting downstream immunomodulatory effects in preclinical models29. Importantly, phage specificity enables selective elimination of pathogenic C. acnes while preserving beneficial cutaneous commensals contributing to skin homeostasis.
Phage-derived endolysins provide an additional therapeutic strategy. These enzymes rapidly degrade peptidoglycan upon contact and do not depend on bacterial metabolic activity. Recombinant and engineered endolysins derived from C. acnes phages show enhanced stability and broad activity across inflammatory subtypes. They also maintain compatibility with lipid-rich skin environments, making them suitable candidates for topical acne formulations30.
Despite encouraging preclinical evidence, several trans- lational challenges remain. The pilosebaceous unit's anaerobic and lipid-density environment can limit phage stability and tissue penetration. To address this and to enhance follicular targeting and sustained release, recent studies have explored delivery approaches including microencapsulation, liposomal carriers, hydrogels, and microneedle-assisted systems31. Add- itionally, individual phages' narrow host range may necessitate use of personalized phage panels or cocktails to ensure diverse clinical strains' coverage19.
Overall, phage and endolysin-based therapies are becoming precision tools for restoring microbial balance, disrupting bio- films, and reducing inflammation in acne vulgaris. For suc- cessful clinical translation, continued advances in formulation technologies, engineered lysins, and strain-level diagnostics are critical32.
2. Bacteriophage and endolysin therapy in atopic dermatitis (AD)
AD is a chronic inflammatory skin disease marked by epidermal barrier dysfunction, immune dysregulation, and pronounced microbial dysbiosis. A hallmark of AD is excessive colonization by S. aureus, closely correlated with disease severity. By releasing proteases, toxins, and superantigens, this pathogen promotes Th2-skewed immune responses, pruritus, and barrier damage3. In parallel, protective commensal staphylococci, such as S. epidermidis and S. hominis, are markedly reduced. Loss of these beneficial species weakens antimicrobial defense and facilitates pathogen dominance33. This shift from a diverse microbial ecosystem to S. aureus-driven dysbiosis represents a central therapeutic AD target.
To correct this imbalance, phages and phage-derived endolysins provide precision-based strategies. Staphylococcal endolysins, particularly those containing CHAP catalytic domains, rapidly cleave peptidoglycans with high specificity. These enzymes demonstrate potent lytic activity against S. aureus, including methicillin-resistant strains, while largely sparing commensal bacteria that support immune homeo- stasis21,23. Clinical studies evaluating the topical endolysin staphefekt SA.100 have shown reductions in S. aureus colonization and improvements in eczema severity scores reported in small clinical studies and case series. Importantly, during prolonged use, these benefits were observed without evidence of resistance development or disruption of beneficial skin microbiota34.
Lytic phages also show therapeutic promise in AD. S. aureus phages can penetrate biofilms, replicate at infection sites, and selectively reduce pathogen burden. Their capacity to degrade extracellular biofilm matrices provides a clear advantage over conventional antibiotics that often show limited efficacy against biofilm-associated bacteria in AD lesions24. In pre- clinical models, phage treatment reduces skin inflammation, lowers bacterial load, and enhances pathways associated with barrier repair, suggesting coordinated antimicrobial and immunomodulatory effects25.
Recent metagenomic analyses further highlight skin virome's role in AD. Significant alterations in phage abundance and diversity have been reported, with numerous viral genomes fluctuating in comparison to those found in healthy skin15. These findings suggest that rather than merely reflecting inflammation, phage-bacterial interactions may actively shape disease severity.
Key translational challenges remain, including formulation stability on inflamed skin and strain heterogeneity among S. aureus isolates. Advances in engineered lysins, patient-specific phage cocktails, and combination approaches with moistur- izers or anti-inflammatory agents may help overcome these barriers35,36. Collectively, phage- and endolysin-based ther- apies represent next-generation tools for restoring microbial balance and improving clinical AD outcomes. However, large randomized controlled trials remain limited, and standardized endpoints for microbiome-directed therapies are still evolving.
3. Phage and endolysin therapy for chronic wounds and polymicrobial skin infections
Chronic wounds represent one of the most challenging settings for antimicrobial intervention. Diabetic foot ulcers, venous leg ulcers, and pressure injuries commonly harbor dense polymicrobial biofilms composed of S. aureus (including MRSA), P. aeruginosa, enterococci, and obligate anaerobes. Biofilm-associated bacteria can tolerate antibiotic concen- trations up to 1,000 times higher than those required to kill planktonic cells, evade host immune clearance, and sustain chronic inflammation, impairing tissue repair and delaying wound healing37,38. Standard treatments, such as debride- ment and broad-spectrum antibiotics, often yield inconsistent outcomes and further contribute to antimicrobial resistance.
In this context, phage therapy offers several advantages. Lytic phages can infect bacteria embedded within biofilms and replicate locally at the infection site. The P. aeruginosa phage PA1Ø is a well-characterized example that utilizes type IV pili as its bacterial receptor and efficiently lyses biofilm-associated cells6. Biofilm-focused experimental studies have demonstrated that PA1Ø reduces bacterial burden and disrupts biofilm architecture, supporting its translational relevance for chronic wound management39.
Endolysins provide a complementary strategy against Gram-positive wound pathogens. The staphylococcal endolysin LysSAP26, derived from phage SAP-26, exhibits potent in vitro lytic activity against MRSA and other clinically relevant pathogens in planktonic and biofilm-associated states23. In contrast, the phage SAP-26 has been shown to display strong antibiofilm activity and marked synergistic effects when com- bined with rifampicin in in-vitro MRSA biofilm assays, resulting in substantial reductions in viable biofilm-associated bacteria compared with antibiotic treatment alone22. Other CHAP-domain endolysins also exhibit broad activity against Gram-positive wound isolates and remain stable across a range of pH conditions, supporting their incorporation into topical hydrogels and wound dressings40.
An additional advantage of phage-based therapy is localized self-amplification. Applied phages replicate only in the pre- sence of susceptible bacteria, increasing their concentration specifically at infected sites while minimizing systemic expo- sure and off-target effects on commensal skin microbiota41. This targeted activity may contribute to microbial rebalancing and reduced inflammatory signaling within chronic wounds.
Despite these benefits, several translational challenges remain. Effective penetration into mature biofilms, strain-level variability among clinical isolates, and integration with existing wound care protocols must be addressed to optimize thera- peutic outcomes. Emerging formulation strategies, including alginate dressings, hydrogel encapsulation, and nanoparticle-based delivery systems, show promise in overcoming these barriers31. Collectively, phages and endolysins represent precision-based therapeutics with significant potential for treating chronic wounds dominated by antibiotic-resistant and biofilm-forming pathogens, warranting further evaluation in clinically relevant wound infection models (Table 2).
|
Skin |
Target |
Therapeutic |
Key mechanism |
Study type |
Outcome |
References |
|
Atopic |
Staphylococcus |
SaGU1
|
Selective adsorption |
In vitro (clinical |
Demonstrated selective |
51
|
|
Atopic |
Staphylococcus |
Staphefekt |
Enzymatic cleavage |
Human case reports |
Reduced S.
aureus |
8, 34 |
|
Acne |
Cutibacterium |
C. acnes |
Highly specific |
In vitro |
Broad lytic activity |
27 |
|
Acne |
Cutibacterium |
CAP10-3 |
Enzymatic |
In vitro assays |
Demonstrated |
52 |
|
Chronic |
Pseudomonas |
PA1Ø |
Type IV pili-dependent |
In vitro and biofilm- |
Effective biofilm |
6, 39 |
|
Chronic |
MRSA |
SAP-26 |
Biofilm penetration |
In vitro MRSA |
Strong antibiofilm |
22 |
|
Chronic |
MRSA, MDR |
LysSAP26 |
Enzymatic |
In vitro antibacterial |
Potent bacteriolytic |
23 |
|
Evidence level reflects the experimental system used
(in vitro, animal models, or human
reports); no agent listed has yet received regulatory approval for
dermatologic indications |
||||||
Phage- and endolysin-based therapeutics represent a pro- mising paradigm shift in management of dysbiosis-driven skin diseases, offering mechanistically distinct advantages over conventional antimicrobial strategies. One of their most significant strengths lies in their high specificity. Lytic bacterio- phages selectively target pathogenic species such as C. acnes, S. aureus, and P. aeruginosa while sparing commensal micro- organisms essential for maintaining skin barrier function and immune homeostasis11. This targeted activity contrasts sharply with broad-spectrum topical and systemic antibiotics, which frequently disrupt microbial diversity, exacerbate dys- biosis, and contribute to disease recurrence and antimicrobial resistance.
Another major advantage of phage- and endolysin-based therapeutics is their efficacy against biofilms, a key factor underlying chronic skin infections and nonhealing wounds' persistence. Biofilm matrices restrict antibiotic penetration and protect bacteria from host immune responses. Multiple lytic phages, including PA1Ø and the staphylococcal phage SAP-26, have demonstrated in experimental models the ability to disrupt established biofilms and eliminate bacteria embedded within mature biofilm structures6,22. Endolysins are particularly effective against Gram-positive pathogens because they in- duce rapid bacterial lysis independent of metabolic state. This growth-phase independence enhances their activity against stationary-phase cells and antibiotic-tolerant populations, including MRSA40.
Phages further possess unique capacity for localized self-amplification. Following topical or local administration, phages replicate exclusively in susceptible bacterial hosts' presence, leading to increased local concentration at infection sites while minimizing systemic exposure and off-target effects on the resident skin microbiota18. This self-limiting pharmacodynamic property may reduce dosing frequency and support sustained antimicrobial activity without cumulative toxicity.
Despite these advantages, several limitations and trans- lational challenges must be addressed. Many phages exhibit a relatively narrow host range, potentially limiting their efficacy against genetically diverse clinical isolates of S. aureus or C. acnes. This challenge may be mitigated using rationally de- signed phage cocktails, strain-matched therapeutic approaches, or adaptive phage formulations19. Formulation stability also remains a concern because phages and endolysins can be sensitive to environmental factors such as pH, temperature, desiccation, UV exposure, and lipid-rich skin environments42. Moreover, regulatory frameworks governing phage-based therapeutics are still evolving, and standardized guidelines across jurisdictions for manufacturing, quality control, genomic safety screening, and clinical evaluation remain limited43.
Emergence of bacterial resistance to phages or endolysins represents another important consideration. However, resist- ance mechanisms differ fundamentally from those associated with antibiotics and might develop more slowly. Importantly, phages retain the ability to coevolve with bacterial populations under selective pressure, potentially restoring therapeutic efficacy in vivo even when resistance is observed in-vitro44. Combination strategies involving phage cocktails, periodic reformulation, or coadministration with antibiotics or endo- lysins may further suppress resistance development.
At this writing, technological and conceptual advances are rapidly expanding phage- and endolysin-based dermato- logical interventions' therapeutic potential. One key direction is personalized phage therapy, enabled by rapid pathogen sequencing, phage susceptibility profiling, and curated phage libraries. This approach is particularly relevant for diseases such as acne vulgaris and atopic dermatitis, in which strain-level diversity and variable phage sensitivity limit uniform treatment strategies' effectiveness36. Indeed, integration of artificial intelligence–assisted host-range prediction tools may further accelerate clinical implementation.
Advances in phage and endolysin engineering are also reshaping the field. Genetically modified phages can be designed to broaden the host range, enhance lytic activity, or interfere with bacterial virulence and quorum-sensing pathways36. Some engineered phages are being explored as delivery platforms for anti-inflammatory or immunomodula- tory molecules, offering dual antimicrobial and host-directed therapeutic effects45. In parallel, engineered endolysins with optimized catalytic domains, enhanced stability, and improved tissue penetration have demonstrated superior activity against drug-resistant Gram-positive pathogens in preclinical studies.
Innovations in drug delivery systems are expected to enhance dermatologic applications further. Microneedle-based plat- forms allow targeted delivery into pilosebaceous units for acne treatment, whereas liposomes, hydrogels, and polymer-based nanoparticles improve stability and enable sustained release on the skin's surface46,47. In chronic wounds, alginate dressings and hydrogel matrices provide a moisture-rich microenvironment that supports phage viability and facilitates biofilm penetration48.
Beyond direct pathogen eradication, phages are increasingly recognized as tools for microbiome engineering. Selective depletion of disease-driving taxa may promote ecological re- balancing rather than indiscriminate microbial suppression. Skin virome studies demonstrating correlations between phage community dynamics and disease activity in AD support this concept15,49. Combining phage therapy with microbiome-supportive interventions, such as probiotics or barrier repair strategies, may further enhance therapeutic durability and clinical outcomes.
In sum, although scientific, regulatory, and formulation challenges remain, precision targeting, antibiofilm activity, microbiome-sparing properties, and the expanding techno- logical toolkit associated with phage- and endolysin-based therapeutics position them as highly promising next-generation approaches for precision dermatologic care.
Microbial dysbiosis and antibiotic-resistant pathogens in- creasingly drive such skin diseases as acne vulgaris, AD, and chronic wound infections. Conventional antimicrobial thera- pies often lack specificity, disrupt beneficial skin microbiota, and show limited efficacy against biofilm-associated bacteria. This review highlights phages and phage-derived endolysins as next-generation, precision-based therapeutics that address these limitations. Phages and endolysins selectively eliminate pathogenic bacteria, penetrate and disrupt biofilms, and preserve commensal microorganisms essential for skin barrier integrity and immune balance. By integrating mechanistic in- sights with disease-specific evidence, this work demonstrates how phage-based strategies can move dermatologic therapy beyond broad microbial suppression toward targeted micro- biome modulation. Advances in phage engineering, endolysin optimization, and topical delivery systems further strengthen their translational potential. Importantly, these biologics offer a promising, scalable investigational approach. Collectively, this study positions phage and endolysin-based approaches as promising investigational tools with the potential to improve management of dysbiosis-driven skin diseases through pre- cision, safety, and ecological restoration.
References
1. Sanchez-Lopez MF, Barrero-Caicedo PA, Olmos-Carval HM, Torres-Medina AF, Alzate-Granados JP. Relationship between skin and gut microbiota dysbiosis and inflam- matory skin diseases in adult patients: A systematic review. The Microbe 2025;7:100342
Google Scholar
2. O'Neill AM, Gallo RL. Host-microbiome interactions and recent progress into understanding the biology of acne vulgaris. Microbiome 2018;6:177
Google Scholar
3. Kong HH, Oh J, Deming C, Conlan S, Grice EA, Beatson MA, et al. Temporal shifts in the skin microbiome asso- ciated with disease flares and treatment in children with atopic dermatitis. Genome Res 2012;22:850-859
Google Scholar
4. Shamsuzzaman M, Kim S, Choi YJ, Kim B. Therapeutic phage candidates for targeting prevalent sequence types of carbapenem-resistant Escherichia coli. Foodborne Pathog Dis 2024;21:681-688
Google Scholar
5. Shamsuzzaman M, Kim S, Kim J. Bacteriophage as a novel therapeutic approach for killing multidrug-resistant Escherichia coli ST131 clone. Front Microbiol 2024;15: 1455710
Google Scholar
6. Kim S, Rahman M, Seol SY, Yoon SS, Kim J. Pseudomonas aeruginosa bacteriophage PA1Ø requires type IV pili for infection and shows broad bactericidal and biofilm removal activities. Appl Environ Microbiol 2012;78:6380-6385
Google Scholar
7. Rimon A, Rakov C, Lerer V, Sheffer-Levi S, Oren SA, Shlomov T, et al. Topical phage therapy in a mouse model of Cutibacterium acnes-induced acne-like lesions. Nat Commun 2023;14:1005
Google Scholar
8. de Wit J, Totté JEE, van Mierlo MM, van Veldhuizen J, van Doorn MB, Schuren FHJ, et al. Endolysin treatment against Staphylococcus aureus in adults with atopic dermatitis: A randomized controlled trial. J Allergy Clin Immunol 2019;144:860-863
Google Scholar
9. Wielscher M, Pfisterer K, Samardzic D, Balsini P, Bangert C, Jäger K, et al. The phageome in normal and inflamed human skin. Sci Adv 2023;9:eadg4015
10. Grice EA, Segre JA. The skin microbiome. Nat Rev Microbiol 2011;9:244-253
11. Belkaid Y, Segre JA. Dialogue between skin microbiota and immunity. Science 2014;346:954-959
Google Scholar
12. Lomholt HB, Kilian M. Population genetic analysis of Propionibacterium acnes identifies a subpopulation and epidemic clones associated with acne. PLoS One 2010; 5:e12277
Google Scholar
13. Kim S, Song H, Lee WJ, Kim J. Antimicrobial susceptibility and characterization of Propionibacterium acnes by multilocus sequence typing and repetitive-sequence-based PCR. Journal of Bacteriology and Virology 2016; 46:135-141
Google Scholar
14. Chng KR, Tay AS, Li C, Ng AH, Wang J, Suri BK, et al. Whole metagenome profiling reveals skin microbiome-dependent susceptibility to atopic dermatitis flare. Nat Microbiol 2016;1:16106
Google Scholar
15. Hannigan GD, Meisel JS, Tyldsley AS, Zheng Q, Hodkinson BP, SanMiguel AJ, et al. The human skin double-stranded DNA virome: Topographical and temporal diversity, genetic enrichment, and dynamic associations with the host microbiome. mBio 2015;6:e01578-15
Google Scholar
16. Wolcott R, Costerton JW, Raoult D, Cutler SJ. The poly- microbial nature of biofilm infection. Clinical Microbiology and Infection 2013;19:107-112
Google Scholar
17. Abedon ST, García P, Mullany P, Aminov R. Editorial: Phage therapy: Past, present and future. Front Microbiol 2017;8:981
Google Scholar
18. Abedon S. Phage therapy pharmacology: Calculating phage dosing. Adv Appl Microbiol 2011;77:1-40
Google Scholar
19. Shamsuzzaman M, Kim S, Kim J. Therapeutic potential of novel phages with antibiotic combinations against ESBL-producing and carbapenem-resistant Escherichia coli. J Glob Antimicrob Resist 2025;43:86-97
Google Scholar
20. Shamsuzzaman M, Choi YJ, Kim S, Kim J. Combating multidrug-resistant uropathogenic E. coli using lytic phages, enhancing antibiotic synergy and inhibiting biofilms. Int Microbiol 2025;28:2749-2761
Google Scholar
21. Choi YJ, Kim S, Dahal RH, Kim J. A novel truncated CHAP modular endolysin, CHAPSAP26-161, that lyses Staphylococcus aureus, Acinetobacter baumannii, and Clostridioides difficile, and exhibits therapeutic effects in a mouse model of A. baumannii infection. J Microbiol Biotechnol 2024;34:1718
Google Scholar
22. Rahman M, Kim S, Kim SM, Seol SY, Kim J. Character- ization of induced Staphylococcus aureus bacteriophage SAP-26 and its anti-biofilm activity with rifampicin. Biofouling 2011;27:1087-1093
Google Scholar
23. Kim S, Jin JS, Choi YJ, Kim J. LysSAP26, a new recom- binant phage endolysin with a broad spectrum anti- bacterial activity. Viruses 2020;12:1340
Google Scholar
24. Sutherland IW, Hughes KA, Skillman LC, Tait K. The interaction of phage and biofilms. FEMS Microbiol Lett 2004;232:1-6
Google Scholar
25. Farfán J, Gonzalez JM, Vives M. The immunomodulatory potential of phage therapy to treat acne: A review on bacterial lysis and immunomodulation. PeerJ 2022;10: e13553
Google Scholar
26. MacNair CR, Rutherford ST, Tan MW. Alternative thera- peutic strategies to treat antibiotic-resistant pathogens. Nature Reviews Microbiology 2023;22:262-275
Google Scholar
27. Marinelli LJ, Fitz-Gibbon S, Hayes C, Bowman C, Inkeles M, Loncaric A, et al. Propionibacterium acnes bacterio- phages display limited genetic diversity and broad killing activity against bacterial skin isolates. mBio 2012;3: e00279-12
Google Scholar
28. Kim S, Song H, Jin JS, Lee WJ, Kim J. Genomic and pheno- typic characterization of Cutibacterium acnes bacterio- phages isolated from acne patients. Antibiotics (Basel) 2022;11:1041
Google Scholar
29. Rimon A, Rakov C, Lerer V, Sheffer-Levi S, Oren SA, Shlomov T, et al. Topical phage therapy in a mouse model of Cutibacterium acnes-induced acne-like lesions. Nat Commun 2023;14:1005
Google Scholar
30. Nakatsuji T, Chen TH, Narala S, Chun KA, Two AM, Yun T, et al. Antimicrobials from human skin commensal bacteria protect against Staphylococcus aureus and are deficient in atopic dermatitis. Sci Transl Med 2017;9: eaah4680
Google Scholar
31. Yan W, Banerjee P, Xu M, Mukhopadhyay S, Ip M, Carrigy NB, et al. Formulation strategies for bacteriophages to target intracellular bacterial pathogens. Adv Drug Deliv Rev 2021:176
Google Scholar
32. Choi YJ, Shamsuzzaman M, Lee JE, Jeon YH, Kim H, Yoon YR, et al. In vivo pharmacokinetics, therapeutic efficacy and immune response of bacteriophage vB_AbaSt_W16 against carbapenem-resistant Acinetobacter baumannii. JAC Antimicrob Resist 2025;7:dlaf121
Google Scholar
33. Nakatsuji T, Chen TH, Butcher AM, Trzoss LL, Nam SJ, Shirakawa KT, et al. A commensal strain of Staphylo- coccus epidermidis protects against skin neoplasia. Sci Adv 2018;4:eaao4502
Google Scholar
34. Totté JEE, van Doorn MB, Pasmans SGMA. Successful treatment of chronic Staphylococcus aureus-related dermatoses with the topical endolysin staphefekt SA.100: A report of 3 cases. Case Rep Dermatol 2017;9:19-25
Google Scholar
35. De Maesschalck V, Gutiérrez D, Paeshuyse J, Lavigne R, Briers Y. Advanced engineering of third-generation lysins and formulation strategies for clinical applications. Crit Rev Microbiol 2020;46:548-564
Google Scholar
36. Shamsuzzaman M, Choi YJ, Kim S, Jeong JY, Hong CA, Kim J. PEGylation overcomes pharmacological barriers to improve systemic pharmacokinetics and therapeutic efficacy of phages against MDR Escherichia coli. J Microbiol Biotechnol 2025;35:e2509050
Google Scholar
37. Bjarnsholt T. The role of bacterial biofilms in chronic infections. APMIS Suppl 2013:1-51
Google Scholar
38. Durand BA, Pouget C, Magnan C, Molle V, Lavigne JP, Dunyach-Remy Cl. Bacterial interactions in the context of chronic wound biofilm: A review. Microorganisms 2022;10:1500
Google Scholar
39. Azeredo J, Sutherland I. The use of phages for the removal of infectious biofilms. Curr Pharm Biotechnol 2008;9:261-266
Google Scholar
40. Schmelcher M, Donovan DM, Loessner MJ. Bacterio- phage endolysins as novel antimicrobials. Future Microbiol 2012;7:1147-1171
Google Scholar
41. Wang Y, Li L, Liang Y, Xu K, Ye Y, He M. Phage therapy for Acinetobacter baumannii infections: A review on advances in classification, applications, and translational roadblocks. Antibiotics 2025;14:1134
Google Scholar
42. Bolsan AC, Sampaio GV, Rodrigues HC, Silva De Souza S, Edwiges T, Celant De Prá M, et al. Phage formulations and delivery strategies: Unleashing the potential against antibiotic-resistant bacteria. Microbiol Res 2024;282: 127662
Google Scholar
43. Verbeken G, Pirnay JP. European regulatory aspects of phage therapy: magistral phage preparations. Curr Opin Virol 2022;52:24-29
Google Scholar
44. Abedon ST. Phage therapy dosing: The problem(s) with multiplicity of infection (MOI). Bacteriophage 2016;6: e1220348
Google Scholar
45. Traore K, Seyer D, Mihajlovski A, Sagona AP. Engineered bacteriophages for therapeutic and diagnostic appli- cations. Dis Model Mech 2025;18:dmm052393
Google Scholar
46. Karimi M, Mirshekari H, Moosavi Basri S, Bahrami S, Moghoofei M, Hamblin MR, et al. Bacteriophages and phage-inspired nanocarriers for targeted delivery of thera- peutic cargos. Adv Drug Deliv Rev 2016;106:45-62
Google Scholar
47. Qu F, Geng R, Liu Y, Zhu J. Advanced nanocarrier- and microneedle-based transdermal drug delivery strategies for skin diseases treatment. Theranostics 2022;12:3372-3406
Google Scholar
48. Pinto AM, Cerqueira MA, Bañobre-Lópes M, Pastrana LM, Sillankorva S. Bacteriophages for chronic wound treatment: From traditional to novel delivery systems. Viruses 2020;12:235
Google Scholar
49. Niedźwiedzka A, Micallef MP, Biazzo M, Podrini C. The role of the skin microbiome in acne: Challenges and future therapeutic opportunities. Int J Mol Sci 2024:25
Google Scholar
50. Gu J, Xu W, Lei L, Huang J, Feng X, Sun C, et al. LysGH15, a novel bacteriophage lysin, protects a murine bacteremia model efficiently against lethal methicillin-resistant Staphylococcus aureus infection. J Clin Microbiol 2011;49:111-117
Google Scholar
51. Shimamori Y, Pramono AK, Kitao T, Suzuki T, Aizawa SI, Kubori T, et al. Isolation and characterization of a novel phage SaGU1 that infects Staphylococcus aureus clinical isolates from patients with atopic dermatitis. Curr Microbiol 2021;78:1267-1276
Google Scholar
52. Kim JI, Hasnain MA, Moon GS. Expression of a recom- binant endolysin from bacteriophage CAP 10-3 with lytic activity against Cutibacterium acnes. Scientific Reports 2023;13:16430
Google Scholar
Congratulatory MessageClick here!