pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed

pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed
Yousif Nazzal Hosee,Mohammed Sami Farhan,Semaa A. Shaban
10.17966/JMI.2025.30.1.1 Epub 2025 April 01
Abstract
Fungal infections pose a significant threat to global health, leading to considerable morbidity and mortality in humans, animals, and plants. The rise of drug-resistant fungal strains, coupled with the limitations of current antifungal therapies, such as toxicity and the potential for resistance, has prompted the exploration of alternative treatments. Medicinal plants, with their bioactive phytochemicals, have emerged as promising sources of antifungal agents. These compounds, which include phenols, alkaloids, terpenoids, and phytosterols, exhibit diverse mechanisms of action, such as disruption of fungal cell membranes, inhibition of cell wall synthesis, and induction of mitochondrial dysfunction. This review discusses the effect of various fungal pathogens, such as Candida albicans, Aspergillus fumigatus, and Fusarium species, highlighting their virulence factors and the diseases they cause. It also examines the antifungal properties of medicinal plants, focusing on specific species and their bioactive compounds targeting fungal cells. This study explores the mechanisms through which phytochemicals such as alkaloids, flavonoids, and saponins inhibit fungal growth and the potential for synergistic effects when combined with conventional antifungals. While acknowledging the importance of safety, standardization, and toxicity evaluations, the review also delves into the role of computational drug-discovery methods, such as molecular docking and artificial intelligence-assisted screening, in identifying potential antifungal candidates. Despite challenges such as the variability in plant composition and the need for further clinical data, this review emphasizes the significant potential of medicinal plants in addressing drug-resistant fungal infections and outlines the directions for future research, including biotechnology, nanotechnology, and the exploration of understudied plant species.
Keywords
Antifungal agents Drug resistance Fungal infections Medicinal plants Phytochemicals
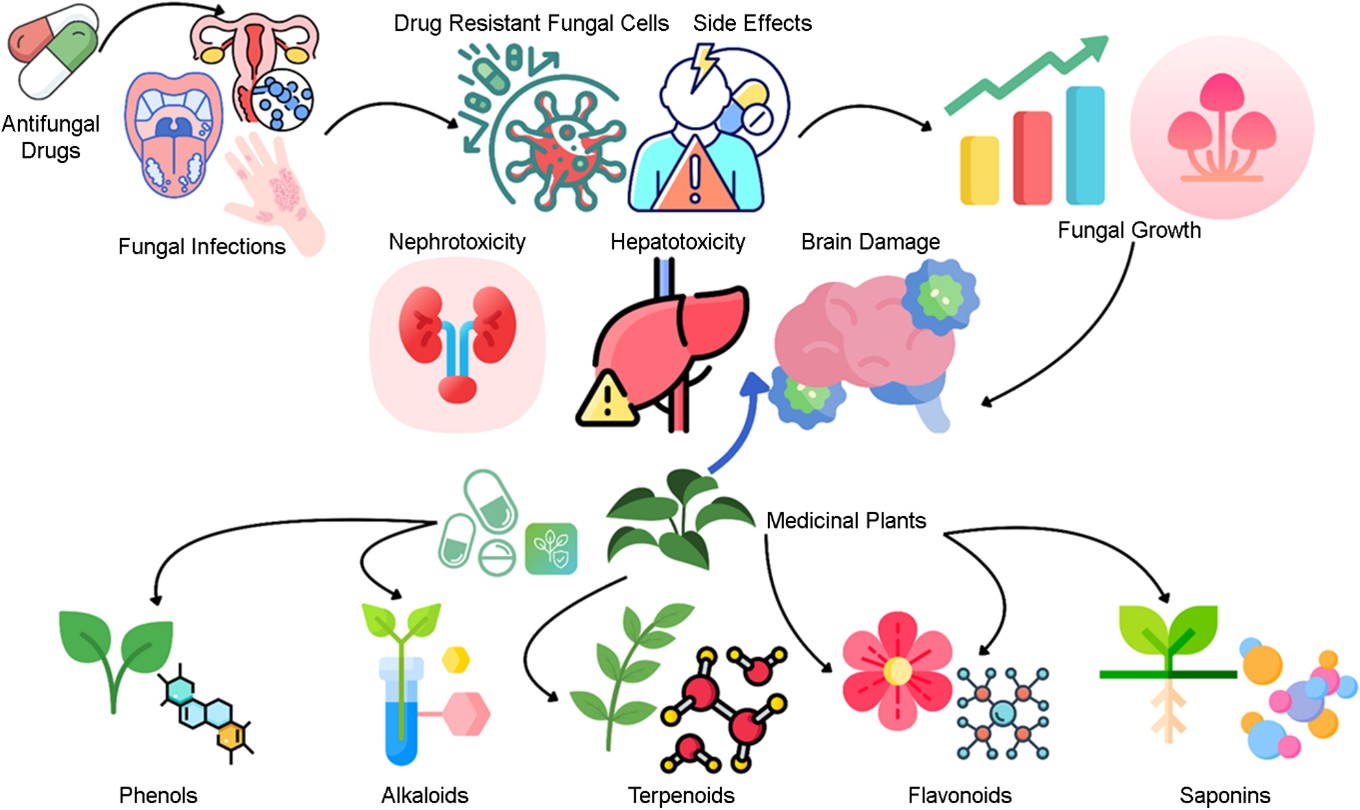
Fungal infections pose a significant challenge for global health, affecting humans, animals, and plants. These infec- tions are a major cause of death and illness, particularly in developing countries. In humans, fungal infections such as invasive candidiasis, aspergillosis, and cryptococcosis account for substantial morbidity and mortality, with millions affected annually and approximately 1.7 million deaths reported. Immunocompromised individuals, including those with HIV/ AIDS, cancer, or diabetes, are especially vulnerable. The long-term use of specific drugs further increases this risk. Fungal infections can target various parts of the body, including the brain, ears, skin, mucous membranes, and internal organs1-4.
In animals, fungal infections can result in respiratory and generalized infections, with tissue predilection varying across species. In plants, fungi are responsible for 70~80% of agri- cultural losses caused by microbial diseases, reducing the nutritional value of crops and producing harmful mycotoxins. Genera such as Fusarium, Aspergillus, and Penicillium are particularly notorious for mycotoxin production, posing risks to food safety and human health5.
The rise of drug-resistant fungal strains exacerbates the global challenge to health. Factors such as the overuse of broad-spectrum antibiotics and the prolonged use of anti- fungal drugs such as fluconazole have contributed to resist- ance. The limited availability of antifungal drugs, coupled with their toxic side effects, emphasizes the urgent need for novel therapeutic approaches6.
There are significant limitations to the use of conventional antifungal treatments, including polyenes, azoles, echino- candins, and flucytosine. These include nephrotoxicity, the development of rapid resistance, drug interactions, and the potential for relapse. Some antifungal medications are asso- ciated with additional side effects, such as hepatotoxicity and visual disturbances, further complicating treatment strategies7.
Medicinal plants are emerging as valuable alternatives and complementary treatments for fungal infections. These plants contain bioactive phytochemicals such as phenols, alkaloids, terpenoids, and phytosterols, which exhibit anti- fungal properties. These compounds act through mechanisms such as damaging fungal cell membranes, inhibiting cell wall and DNA/RNA/protein synthesis, and causing mitochondrial dysfunction. There is a long history of the use of medicinal plants in traditional medicine, with many cultures relying on them for the treatment of various ailments. Plant extracts often combine multiple compounds that work synergistically to suppress fungal growth and reduce the risk of resistance. Moreover, plant-based remedies are generally associated with fewer side effects and improved efficacy8-13.
In this review, we examine the role of medicinal plants and their phytochemicals in combating pathogenic fungi. We also explore plant extracts and isolated compounds as potential sources of novel antifungal agents, along with in silico studies on the molecular interactions of plant compounds with fungal enzymes. Specific plants with demonstrated antifungal pro- perties and their mechanisms of action are highlighted to identify potential therapeutic candidates and support the development of new drugs.
Fungal pathogens are of significant medical importance as they cause a range of infections in humans, with some also affecting animals and plants. These pathogens can be categorized based on the type of infection they cause, such as systemic, dermatophytic, or mucocutaneous infections.
Candida albicans is a common cause of candidiasis, a fungal infection that can affect the skin, mucous membranes, and internal organs. It is a dimorphic commensal, meaning it can exist as a harmless organism but under certain conditions can become pathogenic. Candidiasis is characterized by white patches on the skin and superficial infections, particularly in moist areas of the body. C. albicans is a major source of nosocomial septicemia, acquired in a hospital setting. Other clinically significant Candida species include Candida tropicalis, Candida parapsilosis, and Candida glabrata, which can form biofilms that contribute to antifungal resistance7,12,14.
Dermatophytes, including Trichophyton, Microsporum, and Epidermophyton, cause superficial infections of the skin, hair, and nails (dermatophytoses). In particular, Trichophyton rubrum and Trichophyton mentagrophytes are associated with nail infections (onychomycosis), whereas Microsporum canis and Epidermophyton floccosum are also common der- matophytes. These fungi produce enzymes such as keratinases, proteases, and lipases that enable them to infect keratinized tissues. Dermatophyte infections are especially prevalent in tropical and subtropical areas.
Aspergillus fumigatus is a common cause of aspergillosis, a severe infection that can affect the lungs and other organs, especially in immunocompromised individuals. Some Asper- gillus species, such as Aspergillus niger, produce mycotoxins, including ochratoxin A, which are harmful to both humans and animals15.
Fusarium species can cause infections that range from super- ficial skin and nail infections to invasive diseases, particularly in immunocompromised individuals. These fungi can also cause plant diseases such as head blight and vascular wilt and produce mycotoxins that contaminate food, resulting in to health issues such as cancer and kidney failure.
Mucormycosis, which is caused by fungi in the order Mucorales, including Rhizopus, Mucor, and Lichtheimia species, is a rare but deadly infection primarily affecting immuno- compromised individuals, those with uncontrolled diabetes, and organ transplant recipients. Rhizopus oryzae is the most common cause of mucormycosis, which can affect the brain, lungs, and sinuses. Cryptococcus neoformans is a significant pathogen responsible for cryptococcosis, a systemic fungal infection that affects the lungs and central nervous system, particularly in individuals with HIV/AIDS or other immuno- deficiencies2.
Other fungal pathogens, such as Trichophyton soudanense, Trichophyton vaoundei, Trichophyton erinacei, Microsporum audouinii, and Penicillium species, also play roles in human and plant infections. Penicillium causes blue mold rot in fruits, whereas Botrytis species lead to gray mold in crops, and Pilidiella granati causes heart rot in pomegranates.
These fungal pathogens contribute significantly to human health issues, ranging from superficial skin conditions to life-threatening systemic diseases. The emergence of drug resistance in many of these pathogens highlights the urgent need to develop alternative treatment strategies, including the potential use of medicinal plants.
To establish infections, fungi use various virulence factors. Key enzymes, such as proteases, keratinases, lipases, and phospholipases, facilitate tissue invasion, immune evasion, and inflammation. Proteases, including secreted aspartic pro- teinases (SAPs), assist with fungal morphogenesis and inhibit the immune response by digesting complement components. Keratinases allow dermatophytes to invade the skin, hair, and nails. Lipases and phospholipases hydrolyze host tissues, triggering inflammation. Some fungi, such as Aspergillus and Fusarium, produce mycotoxins such as aflatoxins and trichothecenes, causing severe health issues such as liver damage and cancer16,17.
Other virulence factors include candidalysin, a toxin from Candida species that damages epithelial cells, and hemolysin, which affects red blood cells and phagocytes. Melanin pro- duction helps fungi resist environmental stress. In addition, sulfate transporters, heat shock proteins, and adenosine triphosphate (ATP)-binding cassette transporters may be targeted for antifungal therapies.
The formation of biofilm is a key immune evasion strategy. Biofilms create a protective matrix that shields fungi from antifungal drugs and immune responses, contributing to resistance. C. albicans uses aspartic proteases to evade the complement system, and its ability to switch between yeast and hyphal forms enhances tissue invasion and immune evasion. To develop effective antifungal therapies and combat resistance, understanding these mechanisms is essential. Fig. 1 provides a visualization of the fungal pathogens and their effects.
For millennia, medicinal plants have played a central role in treating fungal infections and other ailments across cultures. In ancient Egypt, herbal remedies were meticulously docu- mented in texts such as the Ebers Papyrus, dating back to 1500 BCE, which included treatments for various conditions. Similarly, more than 5,000 years ago, the Sumerians recorded plant-based remedies, and Traditional Chinese Medicine and Ayurveda in India have long used herbs that contain bioactive compounds such as flavonoids, terpenoids, saponins, alkaloids, and polyphenols. Plant-based treatments have been developed by Indigenous and traditional systems of medicine across the globe, with knowledge often passed down orally through generations9,11,18,19.
Historically, plants have served as effective antimicrobial agents, with many used to treat fungal infections such as ringworm, tinea, scabies, herpes, and eczema. For instance, Calotropis procera has been used to manage skin infections. In many developing countries, medicinal plants remain a primary source of therapy, especially where access to modern health care is limited. Ageratum conyzoides, Asparagus racemosus, Cassia alata, Cassia occidentalis, and Ficus religiosa are ex- amples of plants traditionally used for treating fungal infections. These plants have demonstrated antifungal properties, and others such as Lantana camara and Thymus capitatus have demonstrated antimicrobial activity.
Medicinal plants are rich in secondary metabolites with known therapeutic properties, forming the foundation of modern pharmacologic research. These plants often contain multiple bioactive compounds that work together to inhibit fungal growth through various mechanisms, reducing the likelihood of resistance development. In many cases, they are preferred over conventional antifungal drugs due to their reduced incidence of side effects, better efficacy, and accessibility.
Recent studies have emphasized the potential of medicinal plants as sources of novel antifungal agents. Phytochemicals such as phenolics, alkaloids, terpenoids, and phytosterols found in plants demonstrate antifungal properties. These compounds can disrupt fungal cells, inhibit their growth, and prevent the production of mycotoxin. A. conyzoides, for ex- ample, has demonstrated antifungal action against pathogens such as Aspergillus, Candida, Fusarium, and Phytophthora. Similarly, Syzygium aromaticum and Inula racemosa exhibit significant antifungal activity against Bipolaris oryzae, with compounds such as ursolic acid targeting specific fungal enzymes. Thymus capitatus, known for its antimicrobial pro- perties, has been studied for its pharmaceutical applications, showing potential in treating fungal infections13,21,22.
Plants such as Heliotropium indicum and Grona triflora, which contain compounds such as indicine-N-oxide and quercetin, have demonstrated antifungal activity against C. albicans. Brucea antidysenterica, aloe vera, and Justicia schimperiana also exhibit antifungal effects against C. albicans and A. niger, attributed to alkaloids, flavonoids, and phe- nols. Cinnamomum zeylanicum and its relatives, including Cinnamomum tamala and Trigonella foenum graecum, show antifungal potential through polyphenols such as rutin, kaempferol, and quercetin.
A. racemosus and C. occidentalis have proven effective against dermatophytes such as Microsporum gypseum and Trichophyton mentagrophytes, with active compounds in- cluding saponins and hydroxy anthraquinones. Tristemma mauritianum and Lavigeria macrocarpa demonstrate anti- fungal activity attributed to their phenolic content. C. procera has been shown to inhibit Microsporum and Trichophyton species completely, whereas Vernonia amygdalina has dem- onstrated activity against Botrytis cinerea.
Although the potential of plant-derived antifungal agents is promising, challenges to their full development remain. Current pharmacopeias often lack rigorous validation of plant-based medications, and more research is needed to isolate and characterize active compounds, elucidate their mechanisms of action, and validate their effects in vivo. Standardizing extraction and formulation techniques, assessing chronic toxicologic effects, and exploring interactions with existing medications are essential for ensuring the safety and efficacy of plant-derived treatments.
The synergistic use of plant extracts with conventional antifungals could enhance therapeutic outcomes and offer an attractive alternative for addressing drug resistance. With the continued investigation into novel bioactive compounds, medicinal plants offer valuable sources for discovering new therapeutic agents to tackle the growing problem of drug-resistant fungal infections. Through the expansion of our understanding of these plants and their antifungal properties, we can develop more effective and sustainable treatments for fungal infections in the future.
Phytochemicals are bioactive plant compounds with anti- fungal properties, encompassing groups such as alkaloids, flavonoids, terpenoids, phenolics, saponins, and tannins. These compounds inhibit fungal growth by targeting cell walls, membranes, and metabolic pathways, offering potential as novel antifungal therapies9,23-27.
Alkaloids, which are nitrogen-containing compounds, are known for their diverse biological activities, including anti- fungal effects. For example, an alkaloid from Datura metel has shown activity against Candida and Aspergillus species, whereas other alkaloids, such as jatrorrhizine from Mahonia aquifolium, exhibit antifungal potential against various fungi. Alkaloids such as β-carboline, haloxylines A and B, and piper- idine from Haloxylon salicornium have also demonstrated antifungal properties. Alkaloids typically block enzyme activity, contributing to their antifungal efficacy. Berberine, which is found in Berberis species, is another alkaloid with antibacterial and antifungal properties that disrupts fungal cell membranes and inhibits growth. Quinine, derived from the Cinchona tree, is mainly known for its antimalarial effects but also exhibits antifungal potential, especially when combined with other therapies. Other alkaloids, such as jatrorrhizine, β-carboline, and haloxylines, inhibit fungal metabolic pathways, including cell wall biosynthesis and enzyme activity, broadening the scope of antifungal treatments.
Flavonoids are phenolic compounds that have antioxidant, anticancer, and antimicrobial properties. They disrupt fungal membranes and bind to cell walls, with compounds such as quercetin, kaempferol, and rutin demonstrating anti- fungal activity. Other flavonoids, such as iso-piscerythrone and piscisoflavones, also demonstrate promising antifungal effects. As a result of their polyphenolic structure, flavonoids interact with fungal membranes, altering their fluidity and permeability, resulting in leakage and cell death. Quercetin, which has been widely studied for its antifungal properties, disrupts fungal membranes and enhances the activity of conventional antifungal drugs, indicating its potential for com- bination therapy. Kaempferol, found in cinnamon, contributes to its antifungal effects through its interaction with fungal membranes. Flavonoids such as isorhamnetin, naringenin, and hesperidin also exhibit antifungal activity by interfering with cell wall synthesis, membrane integrity, and cellular pro- cesses such as RNA/protein synthesis, offering a multitarget approach to combating fungal infections28.
Terpenoids, including terpenes, steroids, and saponins, are another group of compounds with significant antifungal pro- perties, especially via the disruption of fungal cell membranes. For instance, phytol, a diterpene, disrupts fungal membranes, which causes ion leakage and the wilting of hyphae. Eugenol, found in S. aromaticum (clove), disrupts fungal membranes, increasing permeability and causing cell death. Carvacrol, a monoterpenoid from oregano and thyme, also disrupts fungal cell membranes. Other terpenoids such as estragole and panicutine show antifungal effects, likely through their inter- action with fungal membranes. Limonoids, a class of oxygen- ated triterpenes found in citrus fruits, also display antifungal properties by interfering with the functions of fungal cells. Sesquiterpenoids, such as drimenin, drimenol, and polygodial, exhibit potent antifungal effects through the disruption of fungal cell membranes. Trichodermin, which is produced by Trichoderma spp., has shown effectiveness against plant pathogenic fungi, further highlighting the biocontrol potential of terpenoids18.
Phenolic compounds, such as tannins, coumarins, quinones, and stilbenoids, are abundant in plants and possess antifungal activity. Caffeic acid and ellagic acid inhibit the growth of Candida auris by modifying the fungal cell wall. Phenolic com- pounds damage fungal membranes, inhibit electron transport enzymes, and disrupt cell walls, with a correlation between increased oxidation and enhanced antifungal potency29,30.
Saponins, which are glycosides with surface-active pro- perties, exhibit diverse biological activities, including antifungal effects. CAY-1, a triterpene saponin from Capsicum frutescens (chili pepper), is potent against various fungal strains, likely due to its ability to disrupt fungal cell membranes. Tannins, which are known for their astringent properties, also exhibit antifungal activity by binding to proteins and inhibiting their synthesis, contributing to the antimicrobial effects of plants such as Ficus vasta and Bersama abyssinica.
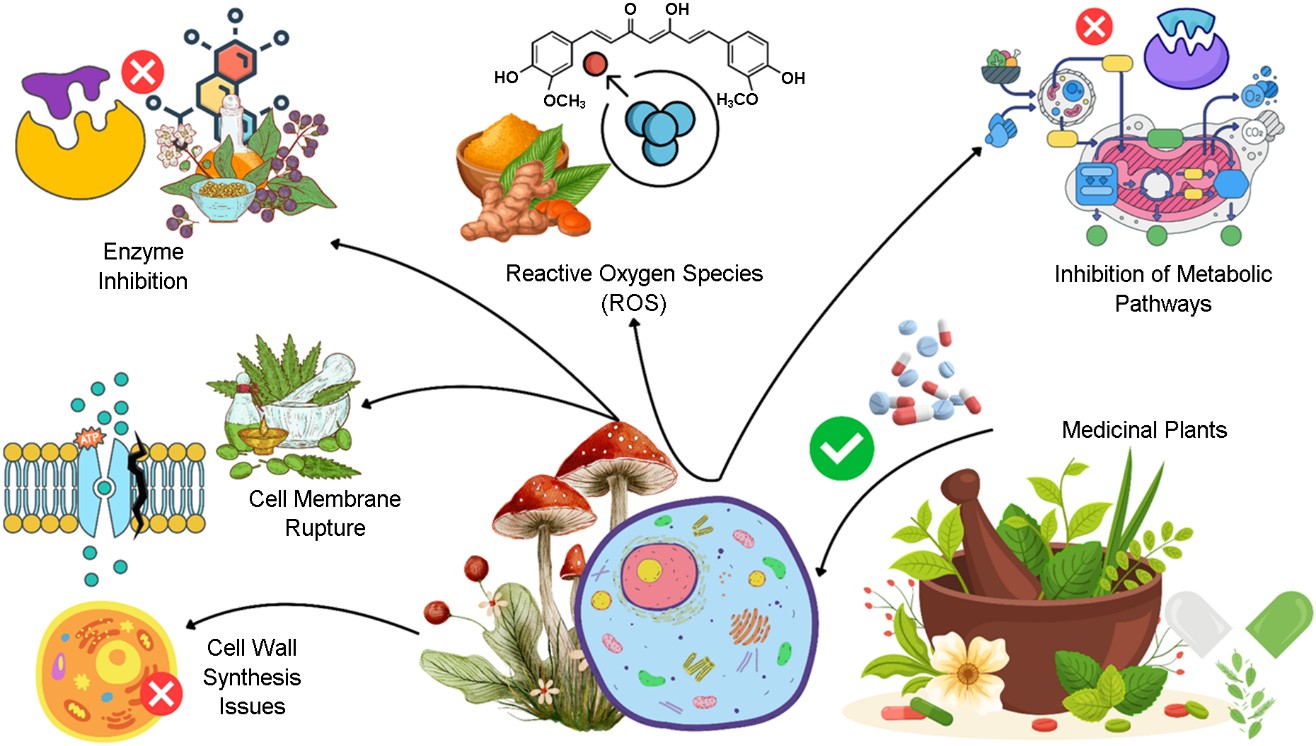
Bioavailability plays a crucial role in the effectiveness of phytochemicals. Techniques such as nanoencapsulation en- hance their absorption and bioavailability, ensuring they are more effective in combating fungal infections. Phytochemicals exert their antifungal effects through mechanisms such as membrane disruption, inhibition of cell wall synthesis, mito- chondrial interference, and the production of reactive oxygen species (ROS). However, variations in the specific phytoche- mical composition of plants, which depend on factors such as geographical location and methods of extraction, can influence their antifungal activity32.
One primary mechanism of antifungal action is the disrup- tion of the cell membrane, as many phytochemicals interact with ergosterol, a crucial component for membrane integrity. Polyene antifungals bind to ergosterol, increasing membrane permeability and causing leakage of cell contents. Saponins also disrupt membrane integrity, leading to cell lysis, whereas flavonoids can alter membrane structure, cause mitochondrial malfunction, and interfere with cellular processes such as cell wall construction and division. Phenolic compounds further damage fungal membranes, inhibit electron transport enzymes, and compromise cell function.
The inhibition of cell wall synthesis is another key antifungal mechanism. Phytochemicals such as flavonoids, phenolic compounds, and certain alkaloids weaken the walls of fungal cells, preventing fungal growth and proliferation. In addition, some phytochemicals interfere with fungal metabolic path- ways, targeting enzymes such as N-myristoyl transferase (NMT) and cytochrome P450 14α-demethylase, which are essential for fungal metabolism. Compounds such as myristicin and rutin hinder the synthesis of critical components such as DNA, RNA, and proteins. Fig. 1 shows the types of phyto- chemicals in medicinal plants.
Phytochemicals derived from medicinal plants can disrupt the walls of fungal cells and membranes through various mechanisms, ultimately inhibiting fungal growth and pre- venting infections. These compounds target key components and processes that are essential for the integrity of the fungal cell wall, membrane stability, and metabolic function33.
Echinocandins, semisynthetic cyclic lipopeptides, block the formation of cell walls by inhibiting the (1,3)-β-D-glucan synthase complex, an enzyme on which fungi rely but that is absent in humans, ensuring their safety for therapeutic use. Other plant extracts inhibit the synthesis of chitin and glucan, which are vital components of the fungal cell wall. Some compounds, such as coumarins, damage fungal cells through the formation of pores in the cell wall, causing cyto- plasmic leakage and necrosis. Meanwhile, saponins disrupt cell membranes, increasing wall permeability. Phenolic com- pounds, along with other phytochemicals, can also damage the cell walls and alter fungal structures. For instance, V. amygdalina extract causes the twisting and folding of fungal mycelia, creating jagged edges and shrinking conidia34.
Certain plant extracts directly target the cell membrane, which has an indirect effect on cell wall function. For example, the seed extract of Cassia fistula damages the membrane of C. albicans, harming yeast cells. Extracts from Piper nigrum (pepper) containing limonene, sabinene, and β-caryophyllene disrupt the fungal cell walls and membranes, coagulating the cytoplasm and damaging organelles. Chitosan also plays a role via the inhibition of gene expression crucial for main- taining cell wall integrity. In addition, flavonoids such as iso-piscerythrone, piscisoflavones, quercetin, and hesperidin show antifungal activity by binding to fungal cell walls. Some plant extracts can even prevent fungal growth by inhibiting the formation of germ tubes, which are essential for hyphal devel- opment35-38.
Phytochemicals can also disrupt the membranes of fungal cells, compromising their function. Many antifungal com- pounds, including polyenes, target ergosterol, a key fungal sterol, reducing membrane fluidity and causing pore formation, ultimately leading to cell death. Eugenol abolishes membrane permeability, resulting in electrolyte loss, whereas saponins perforate lipid bilayers, increasing permeability. Lipophilic flavonoids and terpenoids, such as β-caryophyllene, also dis- rupt membranes. Other compounds, such as sesquiterpenes, reduce ATP reserves and intracellular pH, contributing to cell death. In addition, certain compounds such as silymarin increase oxidative stress and membrane permeability, whereas others such as coumarins and specific terpenoids cause structural changes in the membrane39.
Alkaloids, which are another class of phytochemicals, can block enzyme activity, disrupting essential cellular processes. For instance, myristicin inhibits fatty acid translocation, which is vital for membrane modification. High concentrations of squalene disturb fungal membranes and impair essential func- tions, leading to cell death via mechanisms such as apoptosis, necrosis, or autophagy. These compounds' effectiveness de- pends on the fungal species, compound concentration, and environmental conditions. Plant extracts often contain multiple antifungal compounds, which can have synergistic effects, enhancing their overall efficacy.
Phytochemicals can also target and inhibit fungal enzymes critical for growth and survival, such as chitinase and proteases. The ethanolic extract of F. vasta, for example, has demon- strated strong inhibition of α-glucosidase and α-amylase, outperforming synthetic drugs such as acarbose. Fungal proteases, such as the SAPs (SAP1, SAP2, and SAP3), are virulence factors that contribute to fungal pathogenicity by aiding in adhesion and damaging host tissues. Some plant extracts inhibit fungal growth by preventing the formation of germ tubes, an essential step for hyphal development. For instance, curcumin modulates proteolytic enzyme activity in C. albicans, enhancing antifungal effects.
Phytochemicals also inhibit enzymes involved in ergosterol biosynthesis, a critical pathway for fungal growth. Compounds such as rutin, quercetin, and myristicin bind to fungal enzymes such as 14-alpha demethylase (CYP51), inhibiting ergosterol synthesis and affecting membrane integrity. Other compounds inhibit enzymes such as squalene epoxidase, which is involved in the same pathway. Furthermore, compounds such as α-copaene, cis-calamenene, β-caryophyllene, and caryophyllene oxide inhibit NMT, an enzyme involved in membrane fatty acid translocation36,40.
Some phytochemicals can even block fungal enzymes by binding to their active sites, either competitively or allosterically. For instance, phenolic compounds can oxidize into quinones, inactivating fungal enzymes, whereas alkaloids from B. anti- dysenterica exhibit antimicrobial effects by blocking enzyme activity. Flavonoids also contribute to antifungal activity via the disruption of plasma membranes, inducing mitochondrial dysfunction, and by inhibiting cell division, wall construction, RNA, and protein synthesis. By targeting these enzymes, plant-derived compounds disrupt critical fungal metabolic processes, reducing fungal growth and survival.
Beyond the direct inhibition of fungal growth, phytoche- micals can enhance the immune response of the host. They act as immunomodulators, stimulating the immune system to better combat fungal infections. In addition, by scavenging free radicals, antioxidants in plants help protect the immune system from oxidative stress, thus supporting the integrity of cell membranes and macromolecules. Certain vitamins, such as vitamin D, regulate biochemical pathways that are essential for immune function. Phytochemicals may also help overcome antifungal resistance. Some plant extracts have demonstrated synergistic effects with conventional antifungal drugs, enhancing their efficacy. For example, by boosting ROS production, curcumin has been shown to increase the effects of itraconazole, co-trimoxazole, and amphotericin B. In addition, phytochemicals can disrupt fungal biofilms, which protect fungi from antifungal drugs, and can modulate fungal gene expression associated with resistance mechanisms41-43.
By targeting multiple pathways in fungi, including cell walls, membranes, and metabolic processes, plant-derived com- pounds provide a promising solution for counteracting anti- fungal drug resistance. These compounds not only offer a safe, low-toxicity alternative to synthetic antifungal medications but also hold potential for preventing fungal growth and mycotoxin contamination in food. However, to fully under- stand their mechanisms and optimize their clinical use, further research is needed. Fig. 2, Table 1 provides a visualization of these mechanisms.
|
Phytochemical |
Mechanism
of action |
Pathogen
(s) |
|
Rutin |
Inhibition
of fungal 14-alpha demethylase (CYP51) |
Various
fungal pathogens |
|
Kaempferol |
Inhibition
of fungal 14-alpha demethylase (CYP51) |
Various
fungal pathogens |
|
Quercetin |
May
inhibit fungal growth through various mechanisms |
Candida
albicans
|
|
Indicine-N-oxide |
Binds
to the active pockets of target protein Candidapepsin2 (PDB ID: 3PVK) and
forms |
Candida
albicans |
|
Saponins |
Antifungal
activity against dermatophytes |
Microsporum gypseum, Microsporum |
|
Hydroxy |
Antifungal
activity against dermatophytes |
M. gypseum, M. nanum, |
|
Ellagic acid |
Modifies
the fungal cell wall |
C. auris |
|
Caffeic acid |
Modifies
the fungal cell wall |
C. auris |
|
Curcumin |
May
enhance reactive oxygen species production; |
C. albicans, various fungal |
|
Myricetin |
Exhibits
antifungal effects |
C. albicans, Saccharomyces
cerevisiae |
|
Naringenin |
Exhibits
antifungal effects |
C. albicans, S. cerevisiae |
|
3-O-methylviridicatin |
Antifungal
activity |
Alternaria brassicae, Botrytis cinerea, |
|
Viridicatol |
Antifungal
activity |
A. brassicae, A. alternata, |
|
Fusaripeptide A |
Antifungal
activity |
C. glabrata, C. albicans |
|
Trichothecinol A |
Antifungal
activity |
Cryptococcus albidus |
|
Cytochalasin Z28 |
Fungicidal
activity |
Gaeumannomyces
saubinetti |
|
2-amino-3 |
4-dihydroxy-2-25-(hydroxymethyl)-14-oxo-6 |
12-eicosenoic
acid |
|
Myriocin |
Antifungal
activity |
Cryptococcus
neoformans, |
|
Botryosphaerin H |
Antifungal and nematicidal |
Gaeumannomyces
graminis, Fusarium |
Combination therapy that involves the use of medicinal plant extracts alongside conventional antifungal drugs offers a promising strategy for combating drug resistance and improving treatment outcomes. This approach leverages the synergistic effects of plant-derived compounds and conven- tional antifungals, making it more challenging for fungi to develop resistance and enhancing the overall efficacy of treatment25,27,44.
Numerous studies have demonstrated that combining plant extracts with conventional antifungals results in synergistic effects, in which the combined action is greater than the sum of individual effects. These combinations can enhance the effectiveness of antifungal treatments and potentially overcome resistance mechanisms. For example, extracts of T. mauritianum, L. macrocarpa, and Crassocephalum boughe- yanum showed synergistic effects with ketoconazole against drug-resistant Candida species. Similarly, L. macrocarpa ex- tract showed synergistic effects with both ketoconazole and fluconazole. In addition, Nigella sativa seed extract demon- strated synergistic effects with ciprofloxacin, whereas curcumin exhibited synergistic effects with itraconazole, cotrimoxazole, and amphotericin B, primarily by enhancing ROS production. Furthermore, aqueous extract of Hibiscus sabdariffa combined with antibiotics showed in vitro synergistic effects against Helicobacter pylori45,46.
Other studies have reported additive effects when plant extracts are combined with conventional antifungals. For instance, Murraya koenigii and other plant extracts showed synergistic antiplasmodial activity when combined. In addition, combinations of Carica papaya and V. amygdalina demon- strated synergistic effects in reducing the Plasmodium parasite load in mice. These findings suggest that plant extracts not only have independent antifungal activity but can also enhance the effects of conventional drugs, making them valuable partners in combination therapy.
The mechanisms underlying these synergistic effects are diverse. Plant extracts can disrupt fungal cell membranes, inhibit efflux pumps, and modulate proteolytic enzyme activity. By targeting multiple pathways, plant extracts complicate the development of resistance by fungi. Furthermore, these extracts can enhance the effectiveness of antifungal drugs by increasing drug penetration, inhibiting biofilm formation, and acting as immunomodulators, all of which boost the host's immune response. This multifaceted approach may help combat fungal infections that are difficult to treat using conventional antifungal agents alone47.
Combining different plant-derived compounds often leads to stronger antifungal effects than individual compounds alone as a result of synergistic interactions. This approach enhances the therapeutic potential of plant extracts and helps overcome some of the limitations associated with single-compound treatments. Combining various bioactive molecules found in plants allows for a more robust and comprehensive approach to combating fungal infections. Phytochemicals such as rutin and quercetin, for example, have been shown to enhance the antifungal activity of amphotericin B and fluconazole, whereas curcumin enhances the effectiveness of itraconazole, co-trimoxazole, and amphotericin B.
Moreover, synergistic combinations can help overcome mechanisms of fungal resistance. Fungi often develop re- sistance to antifungal treatments via the alteration of their metabolic pathways or the expulsion of drugs through efflux pumps. By targeting multiple pathways simultaneously, syn- ergistic plant combinations can help reverse or bypass these resistance mechanisms. Some phytochemicals interfere with efflux pumps, disrupt cell wall synthesis, and inhibit biofilm formation, all of which contribute to overcoming drug re- sistance.
In addition to combining plant extracts with conventional drugs, some plant extracts can act synergistically when used together. For example, the combination of M. koenigii leaf, Artocarpus altilis stem bark, A. altilis root, and Enantia chlo- rantha stem bark extracts has exhibited a synergistic effect. Likewise, C. papaya and V. amygdalina, when combined, resulted in enhanced antifungal activity35.
One of the main advantages of synergistic combinations is their broad-spectrum activity. Combining different plant extracts increases the range of fungal species targeted and allows for lower concentrations of individual compounds, reducing the risk of toxicity. This is particularly important because many synthetic antifungal drugs are associated with adverse side effects, whereas plant-based treatments tend to have lower toxicity profiles. The improved efficacy of syner- gistic combinations also results in greater effective control of fungal growth and infections, engaging multiple mechanisms of action to address fungal infections from various angles. By combining compounds with different mechanisms of action, synergistic combinations can delay the development of resistance, reducing the likelihood of fungi adapting to treatment.
From an environmental perspective, plant-based treat- ments are more sustainable and eco-friendly than synthetic drugs are, making them a promising natural alternative to chemical antifungals. However, depending on factors such as the specific plant species, compounds, extraction methods, and the fungal species targeted, the effectiveness of these treatments can vary. Further research is needed to identify optimal combinations and understand their underlying mech- anisms of action.
Standardized extraction and formulation techniques are essential to achieve consistent and reliable results. These techniques help ensure that plant extracts used in combination therapies are potent and effective across different settings.
Although medicinal plants have a long history of use in traditional medicine, their safety and efficacy must be evalu- ated rigorously before they can be widely adopted in modern health care. Although many plant-based remedies have been used for centuries with apparent safety, not all plant-derived compounds are safe. Some plants contain toxic compounds that may cause adverse effects, particularly when used in high doses or for prolonged periods. A significant challenge to the use of medicinal plants is the lack of standardization in their preparation, which can result in variability in the concen- tration of bioactive compounds. Factors such as plant genetics, environmental conditions, and harvesting methods influence the chemical composition of the plant12,48-50.
Certain plant families, such as the Asteraceae family, which includes A. conyzoides, may contain hepatotoxic pyrrolizidine alkaloids (PAs), which can be harmful. However, the presence of PAs varies by region. Before clinical use, it is crucial to conduct thorough toxicologic assessments of natural products and their isolated compounds. Studies on A. conyzoides ex- tracts and compounds suggest they are safe within specific dosing parameters. Mycotoxin contamination also poses a concern, as plant materials with medicinal properties can become contaminated, raising questions about their safety. Herb-drug interactions also require further study to better understand the potential risks of combining medicinal plants with pharmaceutical drugs.
To ensure the safety and effectiveness of plant-derived products, stringent quality control measures are essential. Variations in the concentration of bioactive substances or impurities can affect the reliability and safety of herbal rem- edies. The pharmacokinetics of plant-derived substances, including their absorption, distribution, metabolism, and elim- ination within the body, can also vary greatly. Understanding these properties is essential for developing medications that exhibit consistent and predictable therapeutic effects. To determine the appropriate doses of plant-based medicines to ensure both efficacy and safety, more research is needed. Clinical studies are crucial for validating ethnomedicinal claims and establishing the safety and efficacy of these preparations in humans51.
Some phytochemicals found in medicinal plants can be toxic at high doses or with prolonged use, which makes it important to understand their specific properties and potential risks before they are used. Traditional knowledge accumulated over generations often serves as the foundation for modern drug development. Traditional pharmacopeias, based on the wisdom of native societies, typically rely on plant-based substances rich in secondary metabolites. The ethnopharma- cological approach, which draws from traditional medicine systems, is frequently used to select plants for pharma- cological studies. Due to their higher likelihood of containing bioactive molecules, plants that have a long history of use in traditional medicine are often prioritized for research.
Although traditional use provides a useful starting point, the scientific validation of the efficacy of medicinal plants is necessary through rigorous testing and clinical trials. Modern science aims to identify the active components of medicinal plants and understand their mechanisms of action, which can help determine the most effective and safe ways to use these plants. There is a growing need for clinical trials to confirm the efficacy and safety of plant-derived medicines. These trials are essential for validating the ethnomedicinal claims and determining the safe, effective doses. Modern methods of drug development are also being used to standardize herbal remedies and identify analytical marker biomolecules. Standardized extracts are especially important when the pharmacologic effect results from the synergistic action of multiple compounds.
The World Health Organization has developed policies, guidelines, and strategies to promote the safe use of plant-derived drugs globally. The therapeutic effects of medicinal plants are largely attributed to their chemical constituents, such as secondary metabolites including flavonoids, alkaloids, tannins, saponins, phenols, and glycosides. However, if con- sumed in high doses or for extended periods, some phyto- chemicals can exhibit toxic effects, making it crucial to under- stand their potential toxicity. The concentration of bioactive compounds can vary based on the plant species, growing conditions, and processing methods. In some cases, whole plant extracts may be more effective than isolated compounds, indicating a synergistic effect of multiple compounds.
Using a variety of methods such as virtual screening, mole- cular docking, and molecular modeling, computational drug discovery has revolutionized the identification of potential drug candidates, including antifungal agents. These computational techniques help streamline the drug discovery process by predicting how compounds interact with biological targets, optimizing lead compounds, and assessing drug-likeness early in the development stage9,39,52,53,16,54,53.
Virtual screening is one of the primary techniques used in computational drug discovery. It involves screening large databases of chemical compounds to identify those that may interact with a specific biological target. Virtual screening is often used for antifungal drug discovery to search chemical libraries for compounds that may bind to key fungal enzymes involved in processes such as ergosterol biosynthesis. This is critical for the integrity and growth of the fungal cell mem- brane, and enzymes such as sterol 14α-demethylase (CYP51) play a crucial role in this pathway.
Through virtual screening, researchers can narrow down large chemical libraries and identify potential inhibitors that are likely to interact with the target protein. For example, virtual screening of a dataset of 880 molecules identified more than 600 compounds with good affinity toward CYP51, among which ZINC95486139 and ZINC33833639 stood out as potential inhibitors. These compounds demonstrated favorable druglike properties and high binding affinities, which highlights the potential of virtual screening in dis- covering novel antifungal agents.
By predicting the binding interactions between a ligand (drug candidate) and its target protein, molecular docking further enhances virtual screening. It simulates how the ligand binds to the protein, exploring various orientations and con- formations within the binding site. The binding affinity of each orientation is scored, with compounds that show stronger binding affinity being prioritized for further study. For in- stance, compounds derived from plants such as F. vasta and Dodonaea angustifolia have shown promising docking results with fungal enzymes, such as CYP51 and BMK-13,28,38,55,56.
Docking software, such as AutoDock Vina, PyRx, and CB-Dock2, is commonly used to perform these simulations, and the results provide valuable insights into the nature of ligand-target interactions. These interactions can include hydrogen bonds, hydrophobic interactions, and electrostatic forces, which are essential for ensuring the stability of the ligand-protein complex. The success of docking predictions is often validated by redocking the co-crystallized ligand into the binding site of the target protein. If the docking simulation returns a low root mean square deviation compared with the experimental structure, it indicates that the docking protocol is accurate and reliable.
After identifying potential drug candidates through virtual screening and docking, it is crucial to evaluate their pharmaco- kinetics and toxicity. Absorption, distribution, metabolism, ex- cretion, and toxicity (ADMET) profiling plays an essential role in this evaluation. By predicting how a compound behaves in the human body, ADMET analysis helps researchers identify compounds with favorable druglike properties and those that may exhibit toxicity57.
Tools such as SwissADME are used to assess the pharmaco- kinetics of compounds, including their bioavailability and ability to cross cell membranes. A compound with poor drug-likeness may have poor absorption, which limits its potential as an effective drug. In addition, predicting toxicity early in the discovery process can help eliminate compounds with harmful effects, streamlining the selection of safe and effective drug candidates.
Although molecular docking provides valuable insights into the binding affinity and interactions of compounds, it assumes that the protein-ligand complex is rigid, which may not always be the case in a biological environment. Molecular dynamics (MD) simulations address this limitation by modeling the movement and flexibility of the protein-ligand complex over time. These simulations provide a more realistic view of the stability of the binding interaction and the dynamic behavior of the complex.
MD simulations complement docking studies by confirming the stability of protein-ligand complexes, ensuring that the binding interactions that are observed during docking are maintained over time. Although rigid protein assumptions and computational limitations are challenges, MD simulations remain a powerful tool in the validation of docking pre- dictions and further understanding of the mechanisms of drug action11,55,58,59,59-63.
In addition to traditional computational methods, artificial intelligence (AI) and machine learning (ML) techniques are becoming increasingly important in drug discovery. AI and ML algorithms can analyze vast datasets, identifying patterns and relationships between chemical structures and biological activity. This analysis enables the prediction of novel com- pounds with antifungal activity and the optimization of lead compounds for better efficacy.
Quantitative structure-activity relationship (QSAR) models establish mathematical relationships between a compound's chemical structure and its biological activity. These models are invaluable in the prediction of the antifungal potential of untested compounds based on their structural features. By analyzing chemical descriptors, QSAR models can help researchers design compounds with improved antifungal activity and druglike properties.
Network pharmacology is an emerging field focusing on the interactions between drugs and multiple targets within biological networks. This approach helps improve drug efficacy and reduce side effects by considering the holistic effects of a compound on cellular pathways rather than focusing on a single target. In antifungal drug discovery, network pharma- cology can identify key proteins and pathways involved in fungal growth and survival, guiding the design of multitarget drugs. Fragment-based drug discovery (FBDD) is another pro- mising strategy that identifies small molecular fragments that bind to the target protein. These fragments can be optimized and developed into full-fledged drugs. By targeting enzymes critical for fungal survival, such as CYP51 and NMT, FBDD can be particularly useful in discovering new antifungal agents.
Although progress has been made in computational anti- fungal drug discovery, several challenges must be addressed. Fungal pathogens are eukaryotic organisms that share many similarities with host cells, making it difficult to design selective antifungal agents that do not target human proteins. In addition, fungal strains exhibit genetic diversity, which neces- sitates the development of broad-spectrum agents or com- bination therapies.
Resistance mechanisms, such as mutations in target en- zymes and the formation of biofilms, add complexity to the development of antifungal drugs. Moreover, because many docking protocols treat proteins as rigid structures, the struc- ural flexibility of fungal proteins can result in inaccuracies in docking predictions. In addition, compounds that exhibit good efficacy in vitro may not always perform well in vivo because of issues such as poor pharmacokinetic properties or off-target effects20,64.
Emerging technologies such as AI, deep learning, and multiomics integration offer promising solutions to these challenges. AI and deep learning algorithms can process vast amounts of data, which helps researchers identify patterns and predict antifungal activity more efficiently. Multiomics approaches, which integrate genomics, transcriptomics, pro- teomics, and metabolomics, provide a more comprehensive understanding of fungal biology and reveal new potential drug targets.
Another promising strategy for accelerating drug discovery is drug repurposing. Computational methods can help iden- tify new uses for existing drugs, saving time and resources compared with developing new compounds from scratch. By screening approved drugs for antifungal activity, researchers may uncover new treatments for fungal infections.
Although medicinal plants hold great promise for antifungal drug discovery, their use is accompanied by several challenges and limitations. A primary issue is the variability in plant com- position. The chemical makeup of plant extracts can differ significantly as a result of various factors, including geoeco- logical variations (such as altitude, latitude, and temperature), the specific part of the plant used (whole plant, roots, leaves, etc.), and the timing of sample collection. Environmental factors, including climate, soil composition, and humidity, also influence the ratio of bioactive compounds in plant extracts. In addition, genetic variations within a plant species contribute to this variability. This inconsistency in chemical composition can give rise to unreliable potency and efficacy of plant-based remedies. Furthermore, the presence of multiple compounds in plant extracts may result in synergistic, antagonistic, or unpredictable effects, complicating the development of con- sistent treatments39-42.
The lack of standardization in plant extract preparation and dosages presents another significant challenge. Although standardization is crucial to ensure consistent therapeutic effects, current pharmacopeias often discourage the use of plant-derived medications without established medicinal efficacy. Variations in the concentration of bioactive sub- stances or the presence of impurities can compromise the reliability and safety of herbal remedies. This lack of stand- ardized extraction methods is also a barrier to the use of botanicals in managing plant diseases. The solvent and extraction methods used significantly affect the quality and variety of chemicals with antifungal properties. Moreover, depending on the source plant and the quantities used, the allelopathic effect of botanical fungicides on crops can vary. To ensure the safety and effectiveness of plant-based products, there is an urgent need for standardization and quality control.
Limited clinical data further impedes the widespread adop- tion of medicinal plants for antifungal treatments. Although many plants have been used traditionally for centuries, there is often a lack of scientific evidence to support these claims. To validate ethnomedicinal claims and establish the safety and efficacy of plant-based preparations in humans, clinical studies are essential. Many plant-based remedies are still being researched for their therapeutic potential, but additional in vivo studies are required to evaluate their efficacy in controlled environments. Rigorous clinical trials are necessary to assess their therapeutic potential and safety. Plant extracts often contain complex mixtures of phytochemicals, which can act individually or synergistically, or sometimes antagonistically, leading to varying bioactivities. This complexity highlights the need for more comprehensive studies to fully understand their pharmacologic potential6.
Regulatory and ethical issues also present significant obs- tacles in the development and commercialization of plant-based antifungal agents. The regulation of plant-derived medi- cations is complex and varies by region. Regulatory bodies often require extensive safety and effectiveness testing, which increases the time and cost of the development process. In addition, the commercialization of traditional knowledge and plant resources raises concerns about intellectual property rights and fair compensation for indigenous communities. The overextraction of plant species is a significant concern, as it can lead to resource depletion, threaten plant species, and damage ecosystems. Unsustainable collection practices may also disrupt biodiversity. Ethical considerations must include ensuring the responsible sourcing of plant materials and minimizing the environmental impact of plant harvesting. Furthermore, the safety, toxicity, and quality of plant extracts must be carefully monitored, as some studies have reported mycotoxin contamination or the presence of toxic and muta- genic compounds. The lack of control over herbal drug quality may lead to adverse effects, including toxicity, allergic re- actions, or exposure to harmful bioactive compounds such as cytotoxins or estrogenic agents.
A multidisciplinary approach is needed to address these challenges. To prevent overextraction and preserve biodiversity, conservation initiatives and sustainable harvesting practices are essential. Stringent quality control standards must be established to ensure the safety and effectiveness of plant-based products. Harmonizing regulations across regions would improve the accessibility to plant-derived antifungals, while ethical considerations should ensure that indigenous know- ledge is respected and environmental impacts minimized. Standardizing the extraction and formulation techniques will improve reproducibility and consistency in plant-based antifungal products. Additional research is required to iden- tify promising plants with fungicidal properties, and plant-breeding techniques could enhance the concentration of bioactive compounds in specific species. Identifying the optimal environments for growing certain plant species and develop- ing synthetic analogues of active ingredients could also help maintain quality standards. Regulatory procedures and pricing strategies should be developed to facilitate the timely market introduction of plant-based drugs. To better characterize the chemical composition of medicinal plants, advanced analytical techniques, such as gas chromatography-mass spectrometry and high-performance liquid chromatography, should be used. In silico studies could complement in vivo experiments to better understand the mechanisms of action of plant-derived compounds31,35,36,41. Collaborative efforts between researchers, regulatory agencies, and industry stakeholders are critical in overcoming the challenges associated with medicinal plant-based antifungal drug discovery.
Future research into the discovery of novel antifungal com- pounds from unexplored plant species offers vast potential across multiple areas. A key focus is the identification of phytochemicals as drug leads as well as the application of biotechnology and nanotechnology to enhance plant-based antifungal research.
Many plant species, particularly those from tropical or remote regions, remain understudied and could serve as a rich source of new antifungal compounds. Endophytic fungi and bacteria, which reside within plant tissues, are gaining attention as reservoirs of bioactive compounds, including antifungals. These microorganisms produce metabolites that often mimic those of their host plants, and genetic modifi-cations may enhance the production of desirable compounds. Traditional knowledge of the uses of plants can guide the selection of plants likely to possess antifungal properties, while targeted screening for specific fungal enzymes or pathways may uncover compounds with precise modes of action. A multidisciplinary approach that combines ethnopharmacology, phytochemistry, botany, and analytical chemistry is essential to discovering novel plant-based antifungals. Geographic diversity, especially in regions with neglected tropical diseases, also presents opportunities to uncover new chemical entities.
Several plant compounds have exhibited significant anti- fungal activity, which makes them promising drug leads. Polyphenols, including flavonoids, tannins, and phenolic acids, have shown antifungal, antibacterial, and antiviral properties. For example, rutin, kaempferol, and quercetin have demon- strated the ability to inhibit fungal growth by interacting with fungal proteins. Other bioactive compounds, such as terpe- noids, saponins, anthraquinones, alkaloids, and glycosides, have also demonstrated potential in antifungal therapy. The identification of specific fungal enzymes as targets for plant polyphenols and the exploration of the combined effects of plant-derived chemicals with traditional medications could enhance treatment outcomes and help address drug resistance.
Biotechnology offers promising approaches in enhancing the production of plant-based antifungal agents. Genetic and metabolic engineering of plants or endophytes can increase the synthesis of desired bioactive metabolites. By manipu- lating the biosynthetic pathways of secondary metabolites, researchers can create novel analogs that have improved activity. In vitro culture techniques also allow for testing spe- cific compounds as antifungal agents. Nanotechnology can complement these efforts through improvements in delivery and activity of plant-based compounds. For instance, the plant-mediated synthesis of nanoparticles, such as zinc oxide nanoparticles, can enhance the antifungal activity of plant extracts and offer eco-friendly solutions to control postharvest spoilage. Nanoencapsulation can improve the bioavailability and stability of phytochemicals, protecting them from deg- radation and enhancing their solubility.
Nanoformulations, such as liposomes, polymeric nano- particles, and nanosponges, can also serve as drug-delivery systems. These formulations improve stability, target delivery, and control the release of plant-derived compounds, poten- tially reducing toxicity and enhancing efficacy. Advanced bioinformatics tools, such as molecular docking studies, can predict the binding affinity of bioactive compounds to fungal proteins, aiding in the identification of potent molecules. High-throughput screening methods can rapidly assess the secondary metabolites of plants for their antifungal potential, whereas "omics" technologies, such as genomics and meta- bolomics, offer deeper insights into the genetic pathways responsible for producing bioactive compounds, assisting in uncovering new therapeutic candidates.
Medicinal plants hold significant promise as a source of novel antifungal agents, addressing the urgent challenge of drug-resistant fungal infections. Unlike current antifungal treatments that face limitations such as toxicity and resistance, the bioactive phytochemicals in medicinal plants, including alkaloids, flavonoids, terpenoids, and phenolics, offer diverse and potent mechanisms of action, such as the disruption of fungal cell membranes, inhibition of metabolic pathways, and induction of oxidative stress. In addition, the combination of plant-based compounds with conventional antifungals can enhance treatment efficacy and reduce resistance risks. However, translating these compounds into clinical therapies requires overcoming challenges such as variability in plant composition, lack of standardization, and limited clinical data. Future efforts should focus on improving extraction and formulation techniques, exploring understudied plant species, and leveraging biotechnology to optimize the development of plant-based antifungals. By addressing these barriers, medicinal plants can provide sustainable and effective solutions to combat drug-resistant fungal diseases.
References
1. Kashyap VH, Mishra A, Bordoloi S, Varma A, Joshi NC. Exploring the intersection of Aspergillus fumigatus bio- films, infections, immune response and antifungal re- sistance. Mycoses 2023;66:737-754
Google Scholar
2. Roy M, Karhana S, Shamsuzzaman M, Khan MA. Recent drug development and treatments for fungal infections. Braz J Microbiol 2023;54:1695-1716
Google Scholar
3. Serseg T, Benarous K, Serseg M, Rehman HM, El Bakri Y, Goumri-Said S. Discovery of inhibitors against SARS-CoV-2 associated fungal coinfections via virtual screening, ADMET evaluation, PASS, molecular docking, dynamics and pharmacophore studies. Arab J Basic Appl Sci 2022; 29:337-350
Google Scholar
4. Kashyap VH, Mishra A, Bordoloi S, Varma A, Joshi NC. Exploring the intersection of Aspergillus fumigatus bio- films, infections, immune response and antifungal resist- ance. Mycoses 2023;66:737-754
Google Scholar
5. Auxier B, Debets AJM, Stanford FA, Rhodes J, Becker FM, Reyes Marquez F, et al. The human fungal pathogen Aspergillus fumigatus can produce the highest known number of meiotic crossovers. PLoS Biol 2023;21: e3002278
Google Scholar
6. Zouirech O, Alyousef AA, El Barnossi A, El Moussaoui A, Bourhia M, Salamatullah AM, et al. Phytochemical analysis and antioxidant, antibacterial, and antifungal effects of essential oil of black caraway (Nigella sativa L.) seeds against drug-resistant clinically pathogenic microorganisms. Biomed Res Int 2022;2022:5218950
Google Scholar
7. Araújo GR, Costa PCQGD, Nogueira PL, Alves DDN, Ferreira AR, da Silva PR, et al. In silico and in vitro evaluation of the antifungal activity of a new chromone derivative against Candida spp. BioTech 2024;13:16
Google Scholar
8. Adedokun KA, Imodoye SO, Bello IO, Lanihun AA, Bello IO. Therapeutic potentials of medicinal plants and signifi- cance of computational tools in anti-cancer drug discovery. In: Egbuna C, Rudrapal M, Tijjani H, eds. Phytochemistry, computational tools and databases in drug discovery. Elsevier, Amsterdam; 2023:393-455
Google Scholar
9. Awuchi CG, Akram M, Awuchi CG. Roles of medicinal plants in the diagnosis and treatment of eumycetoma. In: Neglected tropical diseases and phytochemicals in drug discovery. John Wiley & Sons, Ltd; 2021:453-476
Google Scholar
10. Dahiya R, Dahiya S, Shrivastava J, Fuloria NK, Gautam H, Mourya R, et al. Natural cyclic polypeptides as vital phyto- chemical constituents from seeds of selected medicinal plants. Arch Pharm (Weinheim) 2021;354:2000446
Google Scholar
11. Devaraji V, Sivaraman J, Prabhu S. Large-scale computa- tional screening of Indian medicinal plants reveals Cassia angustifolia to be a potentially anti-diabetic. J Biomol Struct Dyn 2024;42:194-210
Google Scholar
12. El-Bondkly EAM, Al Shammari B, El-Gendy MMAA, Alsafari IA, El-Bondkly AAMA, El-Shenawy FS, et al. Phytochemical screening, antifungal, and anticancer activities of medicinal plants Thymelaea hirsuta, Urginea maritima, and Plantago albicans. Biomed Res Int 2022; 2022:9544915
Google Scholar
13. Joshi B, Panda SK, Jouneghani RS, Liu M, Parajuli N, Leyssen P, et al. Antibacterial, antifungal, antiviral, and anthelmintic activities of medicinal plants of Nepal selected based on ethnobotanical evidence. Evid Based Complement Alternat Med 2020;2020:1043471
Google Scholar
14. Chaachouay N, Zidane L. Plant-derived natural products: A source for drug discovery and development. Drugs Drug Candidates 2024;3:184-207
Google Scholar
15. Earle K, Valero C, Conn DP, Vere G, Cook PC, Bromley MJ, et al. Pathogenicity and virulence of Aspergillus fumigatus. Virulence 2023;14:2172264
Google Scholar
16. McEvoy K, Normile TG, Del Poeta M. Antifungal drug development: Targeting the fungal sphingolipid pathway. J Fungi 2020;6:142
Google Scholar
17. Lucio J, Gonzalez-Jimenez I, Roldan A, Amich J, Alcazar-Fuoli L, Mellado E. Importance of the Aspergillus fumi- gatus mismatch repair protein Msh6 in antifungal resist- ance development. J Fungi 2024;10:210
Google Scholar
18. Muthukrishnan S, Prakathi P, Sivakumar T, Thiruvengadam M, Jayaprakash B, Baskar V, et al. Bioactive components and health potential of endophytic micro-fungal diversity in medicinal plants. Antibiotics 2022;11:1533
Google Scholar
19. Ugboko HU, Nwinyi OC, Oranusi SU, Fatoki TH, Omonhinmin CA. Antimicrobial importance of medicinal plants in Nigeria. Sci World J 2020;2020:7059323
Google Scholar
20. Cao Q, Ge C, Wang X, Harvey PJ, Zhang Z, Ma Y, et al. Designing antimicrobial peptides using deep learning and molecular dynamic simulations. Brief Bioinform 2023; 24:bbad058
Google Scholar
21. Zotchev SB. Unlocking the potential of bacterial endo- phytes from medicinal plants for drug discovery. Microb Biotechnol 2024;17:e14382
Google Scholar
22. Lu Y, Zhao J, Cheng L. Virtual screening of antimicrobial agents from medicinal plants. Russ J Bioorganic Chem 2021;47:939-944
Google Scholar
23. Lin YC, Wang CC, Chen IS, Jheng JL, Li JH, Tung CW. TIPdb: a database of anticancer, antiplatelet, and anti- tuberculosis phytochemicals from indigenous plants in Taiwan. Scientific World Journal 2013;2013:736386
Google Scholar
24. Baburam S, Ramasamy S, Shanmugam G, Mathanmohun M. Quorum sensing inhibitory potential and molecular docking studies of Phyllanthus emblica phytochemicals against Pseudomonas aeruginosa. Appl Biochem Bio- technol 2022;194:434-444
Google Scholar
25. Deresa EM, Diriba TF. Phytochemicals as alternative fungi- cides for controlling plant diseases: A comprehensive review of their efficacy, commercial representatives, ad- vantages, challenges for adoption, and possible solutions. Heliyon 2023;9:e13810
Google Scholar
26. Dikkala PK, Kakarlapudi J, Rokalla P, Vedantam SK, Kaur A, Kaur K, et al. Computational screening of phyto- chemicals for anti-diabetic drug discovery. In: Egbuna C, Rudrapal M, Tijjani H, eds. Phytochemistry, computational tools and databases in drug discovery. Elsevier; 2023: 285-311
Google Scholar
27. Nallusamy S, Mannu J, Ravikumar C, Angamuthu K, Nathan B, Nachimuthu K, et al. Shortlisting phytoche- micals exhibiting inhibitory activity against major proteins of SARS-CoV-2 through virtual screening. Reseach Square 2020, doi:10.21203/rs.3.rs-31834/v1surch
Google Scholar
28. Alamri MA, Tahir Ul Qamar M. Network pharmacology based virtual screening of Flavonoids from Dodonea angustifolia and the molecular mechanism against inflam- mation. Saudi Pharm J 2023;31:101802
Google Scholar
29. Khanzada B, Akhtar N, Okla MK, Alamri SA, Al-Hashimi A, Baig MW, et al. Profiling of antifungal activities and In silico studies of natural polyphenols from some plants. Molecules 2021;26:7164
Google Scholar
30. Khatun S, Kim T, Mollah MMI. Heat shock increases the anti-inflammatory and anti-obesity activity of soybean by increasing polyphenol, antioxidant and aglycon form isoflavones. Heliyon 2023;9:e21944
Google Scholar
31. Rahman AU, Abdullah A, Faisal S, Mansour B, Yahya G. Unlocking the therapeutic potential of Nigella sativa extract: phytochemical analysis and revealing antimicro- bial and antioxidant marvels. BMC Complement Med Ther 2024;24:266
Google Scholar
32. Zhu G, Chen S, Zhang Y, Lu L. Mitochondrial membrane-associated protein Mba1 confers antifungal resistance by affecting the production of reactive oxygen species in Aspergillus fumigatus. Antimicrob Agents Chemother 2023;67:e0022523
Google Scholar
33. De Francesco MA. Drug-resistant Aspergillus spp.: A literature review of its resistance mechanisms and its prevalence in Europe. Pathogens 2023;12:1305
Google Scholar
34. Dickwella Widanage MC, Gautam I, Sarkar D, Mentink-Vigier F, Vermaas JV, Ding SY, et al. Adaptative survival of Aspergillus fumigatus to echinocandins arises from cell wall remodeling beyond β-1,3-glucan synthesis in- hibition. Nat Commun 2024;15:6382
Google Scholar
35. Bhairavi VA, Vidya SL, Sathishkumar R. Identification of effective plant extracts against candidiasis: an in silico and in vitro approach. Future J Pharm Sci 2023;9:38
Google Scholar
36. Makhuvele R, Naidu K, Gbashi S, Thipe VC, Adebo OA, Njobeh PB. The use of plant extracts and their phyto- chemicals for control of toxigenic fungi and mycotoxins. Heliyon 2020;6:e05291
Google Scholar
37. Nxumalo KA, Adeyemi JO, Leta TB, Pfukwa TM, Okafor SN, Fawole OA. Antifungal properties and molecular docking of ZnO NPs mediated using medicinal plant extracts. Sci Rep 2024;14:18071
Google Scholar
38. Yabuuchi H, Hayashi K, Shigemoto A, Fujiwara M, Nomura Y, Nakashima M, et al. Virtual screening of anti- microbial plant extracts by machine-learning classification of chemical compounds in semantic space. PLoS One 2023;18:e0285716
Google Scholar
39. Chihomvu P, Ganesan A, Gibbons S, Woollard K, Hayes MA. Phytochemicals in drug discovery—a confluence of tradition and innovation. Int J Mol Sci 2024;25:8792
Google Scholar
40. Nallusamy S, Mannu J, Ravikumar C, Angamuthu K, Nathan B, Nachimuthu K, et al. Exploring phytoche- micals of traditional medicinal plants exhibiting inhibitory activity against main protease, spike glycoprotein, RNA-dependent RNA polymerase and non-structural proteins of SARS-CoV-2 through virtual screening. Front Phar- macol 2021;12:667704
Google Scholar
41. Alemu B, Molla MD, Tezera H, Dekebo A, Asmamaw T. Phytochemical composition and in vitro antioxidant and antimicrobial activities of Bersama abyssinica F. seed extracts. Sci Rep 2024;14:6345
Google Scholar
42. Benoutman A, Erbiai EH, Edderdaki FZ, Cherif EK, Saidi R, Lamrani Z, et al. Phytochemical composition, antioxidant and antifungal activity of Thymus capitatus, a medicinal plant collected from northern Morocco. Antibiotics 2022; 11:681
Google Scholar
43. Kengne IC, Fankam AG, Yamako EK, Tamokou JD. Phytochemical analysis, antifungal, and antioxidant properties of two herbs (Tristemma mauritianum and Crassocephalum bougheyanum) and one tree (Lavigeria macrocarpa) species. Adv Pharmacol Pharm Sci 2023; 2023:2565857
Google Scholar
44. Zhanel GG, Chung P, Adam H, Zelenitsky S, Denisuik A, Schweizer F, et al. Ceftolozane/tazobactam: a novel cephalosporin/β-lactamase inhibitor combination with activity against multidrug-resistant gram-negative bacilli. Drugs 2014;74:31-51
Google Scholar
45. Achilonu CC, Davies A, Kanu OO, Noel CB, Oladele R. Recent advances and future perspectives in mitigating invasive antifungal-resistant pathogen Aspergillus fumi- gatus in Africa. Curr Treat Options Infect Dis 2024;16: 14-33
Google Scholar
46. Azman WN, Hamzah MA, Yahayu M, Zakaria ZA. Molecular docking analysis on antifungal properties of acid condensate produced from palm kernel shell. Environ Qual Manag 2023;33:13-22
Google Scholar
47. Gutierrez-Perez C, Puerner C, Jones JT, Vellanki S, Vesely EM, Xatse MA, et al. Unsaturated fatty acid perturbation combats emerging triazole antifungal resistance in the human fungal pathogen Aspergillus fumigatus. mBio 2024;15:e0116624
Google Scholar
48. Bhat S, Rather M, Gani S, Nabi A, Ganai SA, Shah MD, et al. Identification of plant based potential antifungal compounds against BMK-1 protein of Bipolaris oryzae using molecular docking approach. Sci Rep 2024;14: 15665
Google Scholar
49. Rolta R, Yadav R, Salaria D, Trivedi S, Imran M, Sourirajan A, et al. In silico screening of hundred phytocompounds of ten medicinal plants as potential inhibitors of nucleo- capsid phosphoprotein of COVID-19: an approach to prevent virus assembly. J Biomol Struct Dyn 2021;39: 7017-7034
Google Scholar
50. Savarirajan D, Ramesh VM, Muthaiyan A. In vitro anti- dermatophytic activity of bioactive compounds from selected medicinal plants. J Anal Sci Technol 2021;12: 53
Google Scholar
51. Kurnia D, Lestari S, Mayanti T, Gartika M, Nurdin D. Anti-infection of oral microorganisms from herbal medicine of Piper crocatum Ruiz & Pav. Drug Des Devel Ther 2024;18:2531-2553
Google Scholar
52. Zhou Y, Reynolds TB. Innovations in antifungal drug dis- covery among cell envelope synthesis enzymes through structural insights. J Fungi 2024;10:171
Google Scholar
53. Tiwari P, Bae H. Endophytic fungi: Key insights, emerging prospects, and challenges in natural product drug dis- covery. Microorganisms 2022;10:360
Google Scholar
54. Osset-Trénor P, Pascual-Ahuir A, Proft M. Fungal drug response and antimicrobial resistance. J Fungi 2023;9: 565
Google Scholar
55. Bashir Y, Noor F, Ahmad S, Tariq MH, Qasim M, Tahir Ul Qamar M, et al. Integrated virtual screening and molecular dynamics simulation approaches revealed potential natural inhibitors for DNMT1 as therapeutic solution for triple negative breast cancer. J Biomol Struct Dyn 2024;42:1099-1109
Google Scholar
56. Mteremko D, Chilongola J, Paluch AS, Chacha M. Ensemble-based virtual screening of African natural products to target human thymidylate synthase. J Mol Graph Model 2023;125:108568
Google Scholar
57. Padhi S, Masi M, Chourasia R, Rajashekar Y, Rai AK, Evidente A. ADMET profile and virtual screening of plant and microbial natural metabolites as SARS-CoV-2 S1 glycoprotein receptor binding domain and main protease inhibitors. Eur J Pharmacol 2021;890:173648
Google Scholar
58. Das P, Sercu T, Wadhawan K, Padhi I, Gehrmann S, Cipcigan F, et al. Accelerated antimicrobial discovery via deep generative models and molecular dynamics simula- tions. Nat Biomed Eng 2021;5:613-623
Google Scholar
59. Mishra PC, Alanazi AM, Panda SP, Alam A, Dubey A, Jha SK, et al. Computational exploration of Zika virus RNA-dependent RNA polymerase inhibitors: a promising antiviral drug discovery approach. J Biomol Struct Dyn 2025;43:1689-1700
Google Scholar
60. Rognan D. Molecular dynamics simulations: A tool for drug design. Perspect Drug Discov Des 1998;9:181-209
Google Scholar
61. Childers MC, Daggett V. Insights from molecular dynamics simulations for computational protein design. Mol Syst Des Eng 2017;2:9-33
Google Scholar
62. Wróbel A, Baradyn M, Ratkiewicz A, Drozdowska D. Synthesis, biological activity, and molecular dynamics study of novel series of a trimethoprim analogs as multi-targeted compounds: Dihydrofolate reductase (DHFR) inhibitors and DNA-binding agents. Int J Mol Sci 2021; 22:3685
Google Scholar
63. Yu L, Barros SA, Sun C, Somani S. Cyclic peptide linker design and optimization by molecular dynamics simu- lations. J Chem Inf Model 2023;63:6863-6876
Google Scholar
64. Bennett NR, Coventry B, Goreshnik I, Huang B, Allen A, Vafeados D, et al. Improving de novo protein binder design with deep learning. Nat Commun 2023;14:2625
Google Scholar
Congratulatory MessageClick here!