pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed

pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed
Seung Soo Lee,Seok-Jong Lee,Yong Hyun Jang
10.17966/JMI.2023.28.4.108 Epub 2024 January 05
Abstract
There have been reports of benign and cancerous cutaneous reactions developing at the site of resolved herpes zoster weeks to years after the acute infection. This case study describes a 71-year-old male patient with herpes zoster granulomatous dermatitis, a manifestation of postherpetic isotopic response. His medical history was significant for Graves' disease and chronic myelomonocytic leukemia. After presenting to the emergency department, he was diagnosed with herpes zoster generalisatus. Approximately two months after receiving treatment for herpes zoster, multiple firm subcutaneous nodules appeared on his neck. An excisional biopsy was performed to rule out leukemia cutis. The specimen displayed extensive dermal lymphohistiocyte infiltration, marked by central necrosis and atypical T cells. Tests for tuberculosis and leukemia were negative. At follow-up, an excisional biopsy of the recurrent neck nodules confirmed diffuse dermal infiltration and central necrosis. Based on the clinical and pathological findings, he was diagnosed with herpes zoster granulomatous dermatitis. He was administered systemic and topical steroid therapy over one month, after which the lesions and associated symptoms improved. Wolf's isotopic response refers to the occurrence of a new skin disease at the site of an already healed, unrelated skin disease. This phenomenon is frequently overlooked—especially following herpes zoster. Healthcare practitioners should know Wolf's isotopic response to ensure accurate diagnosis and avoid potential oversight of malignant diseases.
Keywords
Granuloma Herpes zoster Herpes zoster granulomatous dermatitis Wolf's isotopic response
Herpes zoster (HZ) is characterized by unilateral dermatomal pain and a rash. The condition results from the reactivation of latent varicella-zoster virus within neurons. HZ occurs sporadically, with an average incidence ranging from 2 to 5 cases per 1,000 people in community-dwelling populations. The incidence of HZ increases with age, and HZ-related com- plications are also age-associated1. Diminished cell-mediated immunity often affects the elderly, potentially leading to a higher prevalence of comorbidities. Additionally, regardless of age, immunocompromised patients demonstrate an increased risk of complications2.
HZ-related complications are cutaneous—such as dissemi- nated or recurrent zoster—as well as neurological, ophthal- mological, and visceral presentation3. Numerous documented cutaneous disorders emerge at the site of resolved HZ following an acute infection4. This case study presents a case of herpes zoster granulomatous dermatitis (HZ-GD) that developed after disseminated HZ.
A 71-year-old male patient presented to the emergency room with a three-day history of painful erythematous vesicular lesions over his face, neck, trunk, and extremities. Laboratory tests revealed elevated inflammatory markers, including an increased erythrocyte sedimentation rate, leukocyte count, and creatinine. His medical history was significant for Grave's disease; he had been using methimazole for 20 years. Add- itionally, he was recently diagnosed with chronic myelo- monocytic leukemia and undergoing hydroxyurea treatment. A dermatology consultation was requested to manage the skin lesions associated with HZ.
While hospitalized, he received a two-week course of intravenous acyclovir and saline wet dressings. The hydroxy- urea he took to treat his Graves' disease was suspended during this time. His HZ lesions soon improved; however, there was evidence of leukemia progression during follow-up, and the hydroxyurea was reintroduced. Around two months after the HZ lesions were resolved (Fig. 1A-E), he developed firm subcutaneous nodules on his neck, and the HZ lesions reappeared (Fig. 1F).
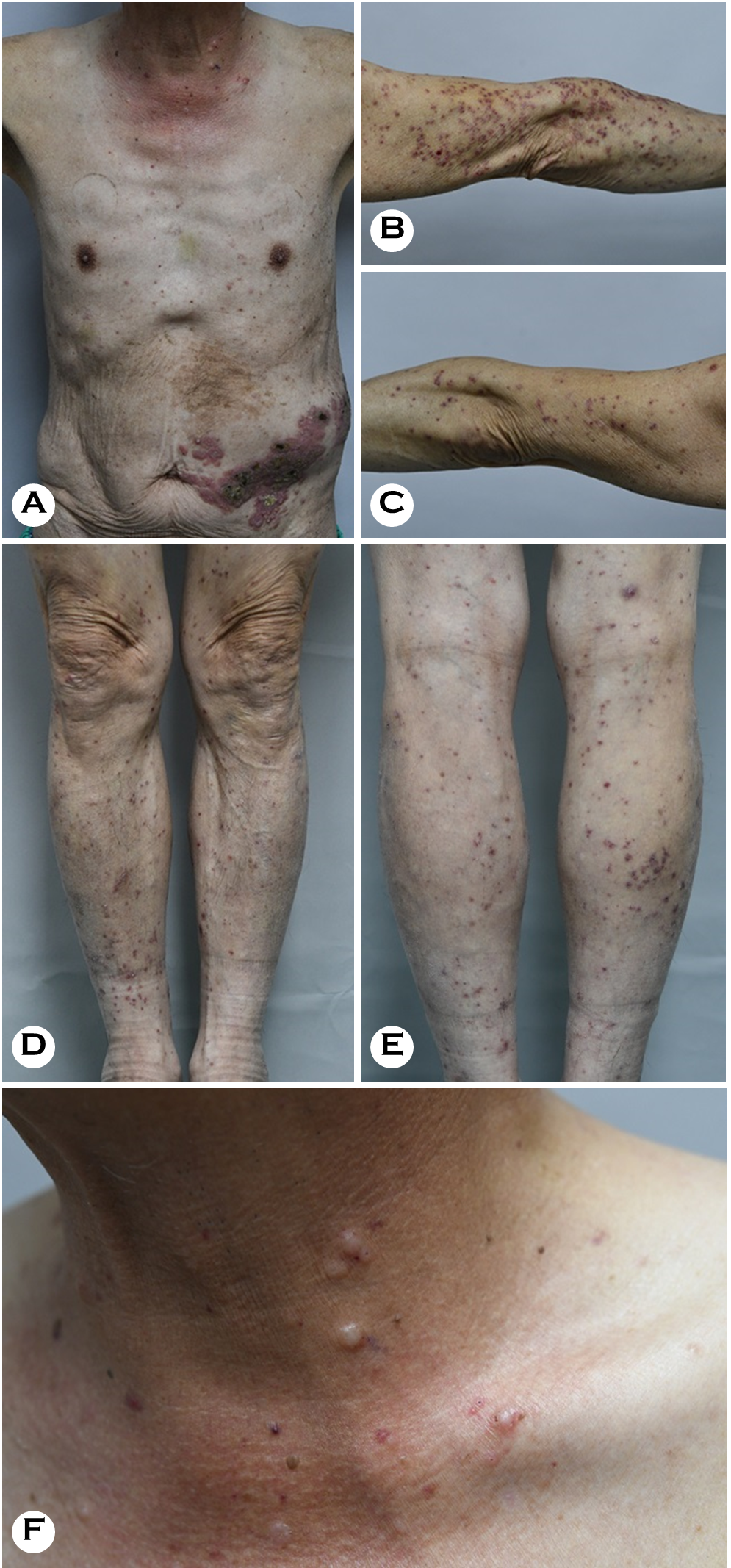
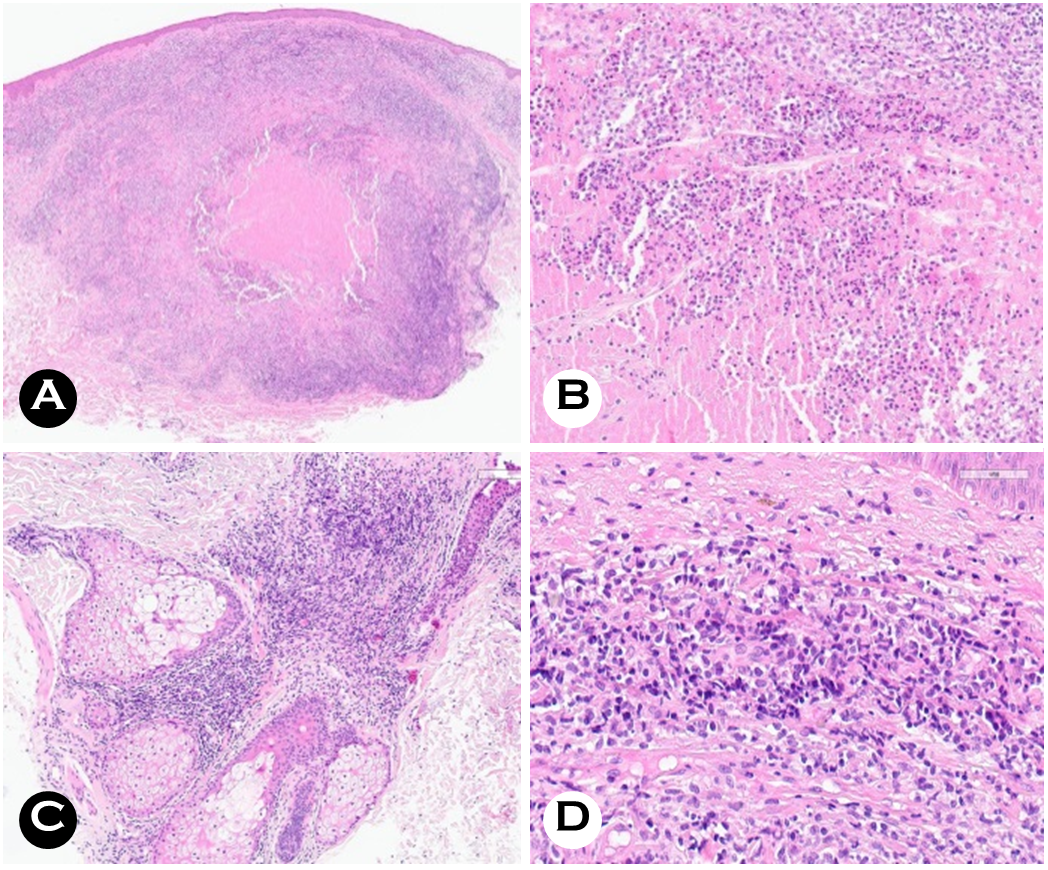
An incisional biopsy was performed on a nodule from the left side of his neck to investigate the possibility of leukemia cutis. Histopathological examination revealed diffuse dermal infiltration of lymphocytes and histiocytes with central necrosis, surrounded by multinucleated giant cells and epithelioid cells (Fig. 2A, B). We observed dense lymphocytic infiltration around the adnexa (Fig. 2C) and atypical T lymphocytes in high-power fields (Fig. 2D). Special staining showed positive results for cluster of differentiation (CD) 3 stain, with Ki-67 levels of 10%. Simultaneously, tests for cytokeratin (CK), CD20, acid-fast bacteria (AFB), and markers indicative of leukemia—such as myeloperoxidase, c-kit, CD34, and CD68—were all negative. Polymerase chain reaction testing for myco- bacterium yielded negative results, excluding tuberculosis as a contributor to the observed caseous granuloma. A chest X-ray revealed left lower lobe peribronchial infiltration, and he was diagnosed with atypical pneumonia and initiated antibiotic therapy. Additional tests were negative, including the interferon-gamma release assay and sputum culture with AFB stain. Evaluation of T-cell receptor beta and gamma gene rearrangement—conducted to investigate the cause of atypical T lymphocytes—revealed polyclonality, thereby excluding lymphoma.
His HZ lesions worsened during follow-up, and he was readmitted to the hospital for a two-week course of intra- venous acyclovir and saline wet dressings. The hydroxyurea prescribed to treat his Graves' disease was stopped while hospitalized. After two weeks of treatment, his dermato- logical symptoms improved (Fig. 3A-C), and hydroxyurea was restarted. However, the emergence of persistent multiple nodules on the face and neck (Fig. 3D-F), along with elevated pinkish patches along the HZ lesions—including the primary site on the left flank—prompted an excisional biopsy.
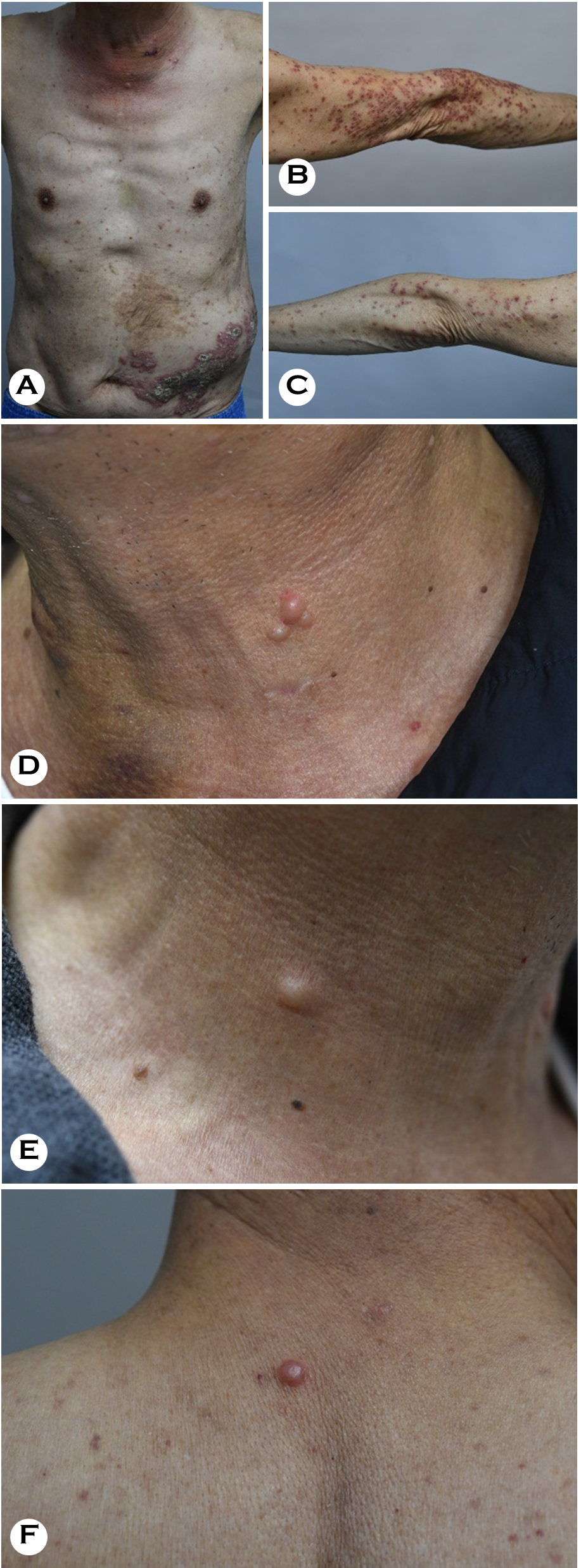
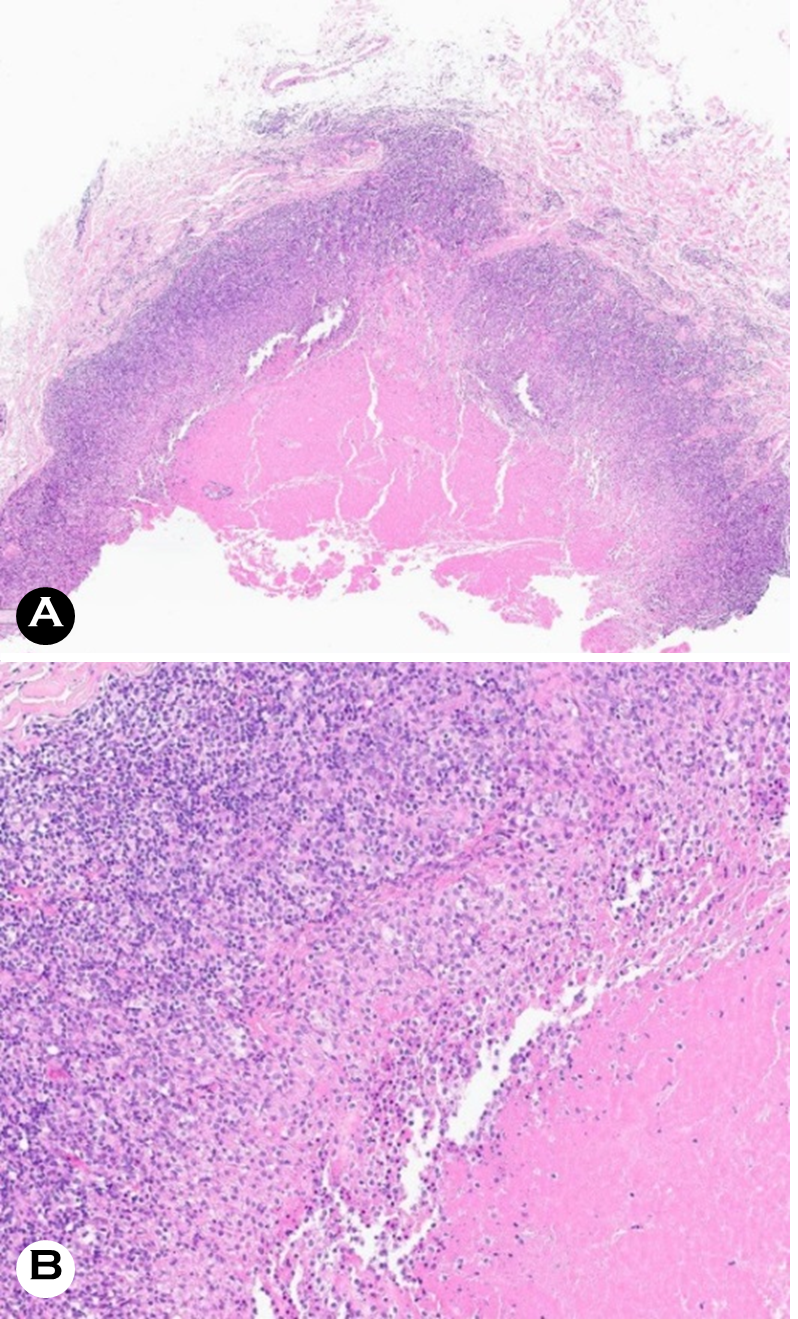
Pathological study revealed a diffuse dermal lymphocytic infiltration with central necrosis surrounded by multinucleated giant cells, epithelioid histiocytes, and perivascular inflam- mation (Fig. 4). Special staining for CD3 was positive, with Ki-67 levels of 10%. At the same time, CK, AFB, c-kit, and CD68 stains were negative. He was diagnosed with granu- lomatous disease based on the characteristic appearance of granulomas featuring central necrosis, surrounded by epithe- lioid histiocytes, lymphocytes, and perivascular as well as peri adnexal inflammation.
Considering the localization of these lesions within the prior HZ site, the patient was conclusively diagnosed with herpes zoster granulomatous dermatitis (HZ-GD). He was treated with topical corticosteroids (desonide 0.05%, des- oximetasone 0.25%), antihistamines, and systemic cortico- steroids (prednisolone 5 mg/day). Within one month, his symptoms were markedly improved.
Wolf's isotopic response refers to the emergence of a new skin condition at a site previously occupied by a different, unrelated skin disease. In contrast, the isomorphic response—known as the Koebner phenomenon and frequently observed in patients with psoriasis and lichen planus—refers to when a skin disorder recurs in areas previously subjected to injury or trauma5,6.
The precise cause of Wolf's isotopic response remains unclear. However, HZ is this phenomenon's most frequently reported precursor condition6. Secondary disorders range from benign to malignant and include granuloma annulare, granulomatous vasculitis, sarcoid granuloma, tuberculoid granuloma, nonspecific granulomatous dermatitis, lichen sclerosus, eosinophilic dermatosis, atypical lymphoid pro- liferation, lymphoplasmacytoid lymphoma, T-cell lymphoma, leukemia cutis, and Kaposi sarcoma4.
Postherpetic isotopic response (PHIR), a manifestation of Wolf's isotopic response, can emerge following an episode of HZ. While rare, PHIR occasionally manifests in immuno- compromised individuals. While its underlying pathogenesis is poorly understood, it is unlikely to be directly induced by HZ, as herpes virus DNA rarely detects skin lesions except during the early stages. Alternatively, PHIR might be linked to local neuroimmune instability resulting from the release of neuropeptides due to damaged cutaneous nerve fibers6. While rare, PHIR encompasses a wide spectrum of secondary disorders ranging from benign to malignant. A granulomatous reaction is the most frequent manifestation in approximately 33.9% of PHIR cases7.
HZ-GD is rare and characterized by localized inflammation, primarily in immunocompromised individuals8. It typically occurs around 4.2 months after HZ onset and is more common in males5. Clinical presentation involves painful or pruritic nodules that follow a distribution similar to that of HZ9. Unlike HZ, vesicles are rarely observed in HZ-GD. Diag- nosis involves evaluating the patient's history of HZ infection, underlying immunocompromising conditions, or medication usage. Clinical assessments should consider characteristic features like lesion distribution, affected areas, and accom- panying symptoms4-7.
Confirmation of HZ-GD typically requires a skin biopsy to identify the presence of granulomas and the clustering of immune cells. Histopathologically, the granulomas appear composed of epithelioid histiocytes, giant cells, and lympho- cytes encircling a central area of necrosis and accompanied by perivascular as well as perineural inflammation4.
There are no established treatment guidelines for HZ-GD; treatment is largely informed by clinical experience and reported cases. Various approaches have been tried, including topical, intralesional, and systemic corticosteroids, which have shown efficacy in many patients5.
There are limited reports about the specific occurrence of Wolf's isotopic response, particularly following HZ. In our case, granulomatous dermatitis has presented as a relatively benign course in the cases we have encountered. However, emphasizing that the spectrum of conditions manifesting as PHIR can encompass a broad range, from benign to malig- nant, is crucial. While this phenomenon is uncommon, PHIR must receive more recognition. This recognition is crucial to facilitate precise diagnoses and prevent any oversight regarding the potential occurrence of malignant diseases.
References
1. Levin MJ, Schmader KE, Oxman MN. Varicella and herpes zoster. In: Kang S, Amagai M, Bruckner AL, Enk AH, Margolis DJ, McMichael AJ, et al., editors. Fitzpatrick's Dermatology, 9th ed. NewYork: McGraw Hill; 2019: 3035-3054
2. Johnson RW, Alvarez-Pasquin MJ, Bijl M, Franco E, Gaillat J, Clara JG, et al. Herpes zoster epidemiology, man- agement, and disease and economic burden in Europe: A multidisciplinary perspective [published correction appears in Ther Adv Vaccines 2016;4:32]. Ther Adv Vaccines 2015;3:109-120
Google Scholar
3. Boivin G, Jovey R, Elliott CT, Patrick DM. Management and prevention of herpes zoster: A Canadian perspective. Can J Infect Dis Med Microbiol 2010;21:45-52
Google Scholar
4. Ferenczi K, Rosenberg AS, McCalmont TH, Kwon EJ, Elenitsas R, Somach SC. Herpes zoster granulomatous dermatitis: histopathologic findings in a case series. J Cutan Pathol 2015;42:739-745
5. Yoon JH, Jang YJ, Park EJ, Kim KJ, Kim KH. A case of herpes zoster granulomatous dermatitis: Report of Wolf's isotopic response. Ann Dermatol 2021;33:186-189
6. Wang T, Zhang M, Zhang Y, Zhang Y, Zhang S, Qu T, et al. Wolf's isotopic response after herpes zoster infection: A study of 24 new cases and literature review. Acta Derm Venereol 2019;99:953-959
Google Scholar
7. Ruocco V, Ruocco E, Brunetti G, Russo T, Gambardella A, Wolf R. Wolf's post-herpetic isotopic response: Infections, tumors, and immune disorders arising on the site of healed herpetic infection. Clin Dermatol 2014;32:561-568
Google Scholar
8. McCoy WH 4th, Otchere E, Musiek AC, Anadkat MJ. Granulomatous dermatitis as a postherpetic isotopic response in immunocompromised patients: A report of 5 cases. JAAD Case Rep 2018;4:752-760
Google Scholar
9. Kuet K, Tiffin N, McDonagh A. An unusual eruption fol- lowing herpes zoster infection. Clin Exp Dermatol 2019; 44:197-199
Congratulatory MessageClick here!