pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed

pISSN : 3058-423X eISSN: 3058-4302
Open Access, Peer-reviewed
Woo Jung Jin,Gyeong Je Cho,Seong Min Hong,Jong Uk Kim,Jung Eun Seol,So Hee Park,Jong Soo Choi,Ki-Hong Kim,Hyojin Kim
10.17966/JMI.2021.26.2.28 Epub 2021 July 01
Abstract
Background: Many studies have investigated onychomycosis in various populations. However, no study has been conducted among individuals living in fishing villages, who seem to be more susceptible to onychomycosis.
Objective: This study was conducted to investigate the prevalence, clinical and mycological characteristics, and risk factors of onychomycosis in individuals living in fishing villages located on two small islands.
Methods: Individuals who had resided in fishing villages located on two small islands in Korea were included. Inspection and a questionnaire survey were performed, and clinical assessment and mycological identification using polymerase chain reaction were performed for those diagnosed with onychomycosis on inspection.
Results: Among the 43 subjects enrolled, 32 (74.4%) were clinically diagnosed with onychomycosis. Causative organisms were identified in 23 (53.5%) subjects, and the most common identified causative strain was Trichophyton rubrum (91.3%), followed by Trichophyton species (4.3%) and Candida species (4.3%). Distal and lateral subungual onychomycosis was the most common clinical type (78.1%), followed by total dystrophic onychomycosis (12.5%). In 10 subjects (31.3%), five or more nails were affected. Among the patients who received treatment for onychomycosis (n = 19), 66.7% had used only conventional topical agents.
Conclusion: Onychomycosis is more prevalent and more severe in people living in fishing villages than in the general population. These findings suggest that environmental or occupational background should be considered in managing onychomycosis.
Keywords
Fishing village Onychomycosis Trichophyton rubrum
Onychomycosis (OM) is a common nail disorder caused by fungi, including dermatophytes, non-dermatophytic molds, and yeasts. Approximately 3~14% of the population is affected by OM. However, due to the influence of environ- mental factors on OM, the degree of risk varies in specific populations based on occupation types or characteristic con- ditions. Accordingly, many studies have investigated OM in various specific populations that were considered to have a higher risk.
We hypothesized that living in a fishing village located on a small island is related to a higher risk of OM. Greater exposure to a humid environment and limited spatiotemporal conditions could make the residents vulnerable to OM. Our pilot study that was conducted on residents of two fishing villages has shown higher rates of clinically diagnosed OM (48/62, 77.4%) and mycologically diagnosed OM (21/28, 75%). With this background, we investigated the prevalence, clinical/mycological features, and risk factors of OM in in- dividuals living in fishing villages located on two small islands.
1. Study design and population
Among the participants in the volunteer medical service provided by Inje University Busan Paik Hospital in 2018 (n = 55), those who agreed to join this study and who have lived in fishing villages located on two islands (Hwa (H) and Isu (I) Islands) located in the southeastern part of Korea (Geoje, Gyeongsangnam-do) were enrolled. This study was approved by the Institutional Review Board of Inje University Busan Paik Hospital (BPIRB 2018-01-123-004), and informed consent was obtained from each subject. All subjects were inspected by two dermatologists and surveyed with a questionnaire. Clinical assessment and mycological evaluation of OM were conducted for those who were diagnosed with OM on inspection.
2. Survey
The subjects were surveyed using a questionnaire designed to collect information on sociodemographic characteristics (age, sex, and occupation), underlying diseases, lifestyle re- garding the wearing of occlusive equipment, behavior for personal hygiene, and treatment history of OM.
3. Clinical assessment
For those who were diagnosed with OM on inspection, the clinical aspects of OM, such as the disease duration, number of involved nails, percentage of total involved area (involved area/total area of the involved nail) that was categorized into three groups (<25%, 25~75%, and >75%), and clinical phenotypes of OM, were assessed.
4. Mycological evaluation
The undersurface of the most prominently involved nail plate was scraped using a scalpel. Samples from each subject were used for polymerase chain reaction-reverse blot hybridi- zation assay (PCR-REBA). In the PCR-REBA test, REBA Fungus-ID® (M&D, Wonju, Korea) was used to examine the color changes of the nucleotide probe based on species-specific internal transcribed spacer sequences.
5. Statistical analysis
Statistical analysis was performed using Statistical Package for the Social Sciences (version 23.0; IBM Corp., Armonk, NY, USA). The chi-square test was performed to evaluate the relationship between wearing occlusive equipment and OM. Behavior for personal hygiene related to OM was analyzed using the independent t-test. Logistic regression analysis was performed to calculate the odds ratio (OR) of wearing occlusive equipment. P-values of less than 0.05 were used to denote statistical significance.
1. Demographic information and point pre- valence of OM
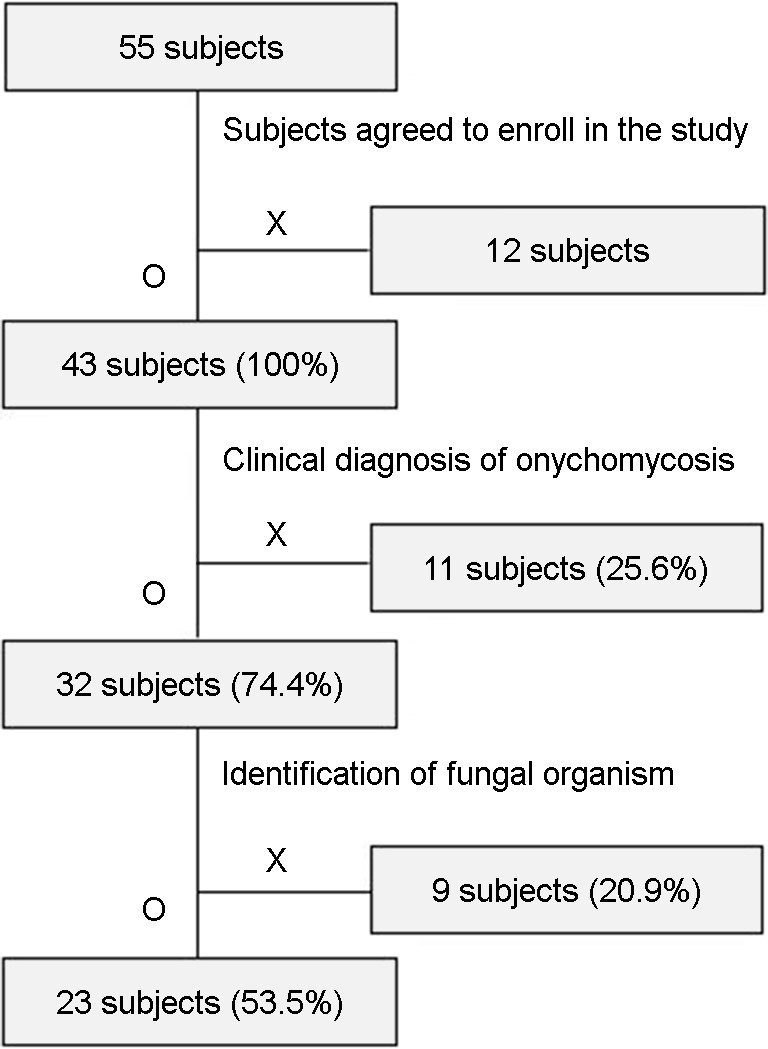
Fifty-five patients participated in the volunteer medical service clinic (Fig. 1). The total number of enrolled subjects was 43 (H = 22 and I = 21). The male-to-female ratio was 1:1.5. Regarding the age groups, 70s was the most common (37.2%), followed by 60s (23.3%) and above 80s (20.9%). Regarding the distribution of occupations, fisherman was the most common (51.2%), followed by farmer (18.6%) (Table 1). Thirty-two subjects were clinically diagnosed with OM on inspection (H = 17 and I = 15). The point prevalence of OM obtained by clinical diagnosis was 74.4% (Table 2).
|
Demographic |
All subjects n (%) |
Clinically n (%) |
|
Sex |
|
|
|
Male |
17 (39.5) |
15 (46.9) |
|
Female |
26 (60.5) |
17 (53.1) |
|
Age
(years old), |
69.9 |
72.0 |
|
<60 |
8 (18.6) |
5 (15.6) |
|
60~69 |
10 (23.3) |
6 (18.8) |
|
70~79 |
16 (37.2) |
13 (40.6) |
|
≥
80 |
9 (20.9) |
8 (25.0) |
|
Occupation |
|
|
|
Fisherman |
22 (51.2) |
16 (50.0) |
|
Farmer |
8 (18.6) |
8 (25.0) |
|
Housewife |
4 (9.3) |
4 (12.5) |
|
Unemployed |
6 (14.0) |
4 (12.5) |
|
Storekeeper |
3 (7.0) |
0 (0.0) |
|
Total |
43 (100.0) |
32 (100.0) |
|
Clinical n (%) |
Mycologically n (%) |
|
|
Hwa Island |
17 (77.3) |
12 (54.5) |
|
Isu Island |
15 (71.4) |
11 (52.4) |
|
Total n (%) |
32 (74.4) |
23 (53.5) |
2. Mycological characteristics of OM
The PCR-REBA test was performed for those who were diagnosed with OM in H and I (n = 32). Twenty-three subjects showed positive results (H = 12 and I = 11), and the point prevalence of OM obtained by clinical and mycological diag- noses was 53.5% (Table 2).
The most common identified causative strain was Tricho- phyton rubrum (91.3%), followed by Trichophyton species (4.3%) and Candida species (4.3%) (Table 3).
|
Causative |
Hwa Island (N=17) n |
Isu Island (N=15) n |
Total (N=32) n |
|
Trichophyton rubrum |
11 |
10 |
21 |
|
Trichophyton species |
0 |
1 |
1 |
|
Candida species |
1 |
0 |
1 |
|
Total n (%) |
12 (70.6) |
11 (73.3) |
23 (71.9) |
3. Clinical characteristics of OM (n = 32)
Most subjects (87.5%) had OM lesions in the toenail. In 62.5% of the subjects, three or more nails were involved (Table 4). The first toenail was the most commonly affected nail (93.8%).
In the analysis of the percentage of the total involved area, 12 subjects (37.5%) showed >75%, and 10 subjects (31.3%) showed 25~75%.
Regarding the clinical type of OM, distal lateral subungual OM (DLSO) was the most common type (78.1%), followed by total dystrophic OM (TDO) (12.5%) and superficial white OM (SWO) (6.3%) (Table 5).
Regarding the disease duration, 21 subjects (65.6%) showed more than 3 years, and four subjects (12.5%) showed 1~3 years.
|
Characteristics |
1~2 n |
3~4 n |
≥ 5 n |
Total n (%) |
|
Fingernail only |
0 |
0 |
0 |
0 (0.0) |
|
Toenail only |
12 |
10 |
6 |
28 (87.5) |
|
Both |
0 |
0 |
4 |
4 (12.5) |
|
Total n (%) |
12 |
10 |
10 |
32 |
|
Characteristics |
DLSO† n |
SWO n |
TDO n |
Combined n |
Total n (%) |
|
Fingernail only |
0 |
0 |
0 |
0 |
0 (0.0) |
|
Toenail only |
22 |
2 |
4 |
0 |
28 (87.5) |
|
Both |
3 |
0 |
0 |
1‡ |
4 (12.5) |
|
Total n (%) |
25 (78.1) |
2 (6.3) |
4 (12.5) |
1 (3.1) |
32 (100.0) |
|
†DLSO: distal and lateral superficial
onychomycosis, SWO:
superficial white onychomycosis, ‡DLSO + TDO type |
|||||
4. Survey results (n = 43)
1) Lifestyle
The results of the analysis of the association between occlu- sive equipment and fingernail/toenail OM are summarized in Table 6. A positive correlation was observed between wearing rubber boots and toenail OM (p = 0.168). Regarding the behavior for personal hygiene, comparisons between subjects with and without OM are summarized in Table 7. The average frequency of taking a shower, washing the hands /feet, and drying increased in subjects with OM; however, no statistical significance was observed (p = 0.577, 0.274, and 0.457, respectively). Logistic regression analysis showed the ORs for wearing rubber boots (2.64; 95% confidence interval (CI), 0.649~10.734; p = 0.175) and gloves (1.85; 95% CI, 0.277~12.389; p = 0.525), and no significant difference was found.
|
Characteristics |
Wearing occlusive gloves (+) n (%) |
Wearing occlusive gloves (-) n (%) |
Total n (%) |
|
Fingernail
onychomycosis (+) |
3 (7.0) |
2 (4.7) |
5 (11.6) |
|
Fingernail
onychomycosis (-) |
21 (48.8) |
17 (39.5) |
38 (88.4) |
|
Total
n (%) |
24 (55.8) |
19 (44.2) |
43 (100.0) |
|
Characteristics |
Wearing rubber boots (+) n (%) |
Wearing rubber boots (-) n (%) |
Total n (%) |
|
Toenail
onychomycosis (+) |
22 (51.2) |
10 (23.3) |
32 (74.4) |
|
Toenail
onychomycosis (-) |
5 (11.6) |
6 (14.0) |
11 (25.6) |
|
Total
n (%) |
27 (62.8) |
16 (37.2) |
43 (100.0) |
|
Characteristics |
Mean frequency of taking n (range) |
Mean frequency of hand/foot washing (per day) n (range) |
Mean frequency of hand/foot drying (per day) n (range) |
|
Onychomycosis
(+) |
5.8
(1~10) |
4.9 (0~10) |
1.0 (0~10) |
|
Onychomycosis
(-) |
5.4
(0~10) |
4.0
(2~7) |
0.5
(0~2) |
|
p-value |
0.577 |
0.274 |
0.457 |
2) Treatment history
Less than half of the enrolled subjects (19/43) reported that they received OM treatment, and topical agent was the most common treatment method used (15/19).
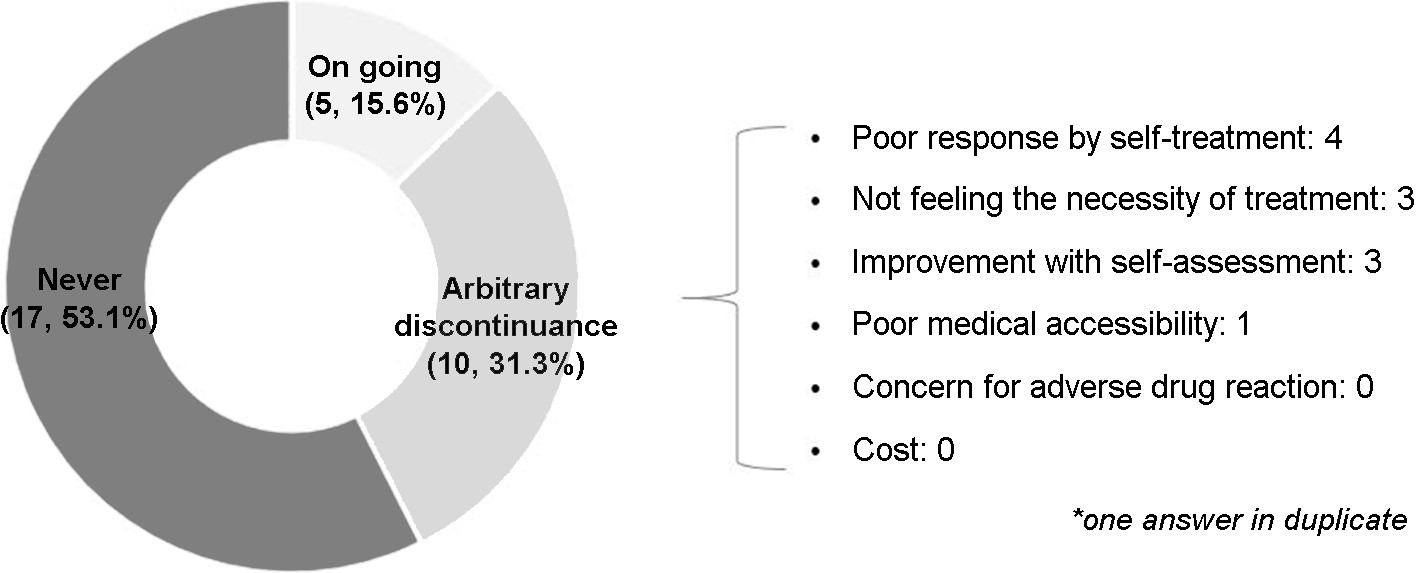
Among subjects with clinically diagnosed OM, less than half (15/32) received OM treatment, and 66.7% of them (10/15) used only topical agents. The reasons for the arbitrary discontinuance of treatment before complete cure were as follows: poor response based on self-assessment, not feeling the necessity of the treatment, improvement with self-assessment, and poor medical accessibility (Fig. 2).
3) Comorbidity
Among subjects with clinically diagnosed OM, 26 (81.3%) had comorbid diseases. The most prevalent comorbidity was hypertension (43.8%), followed by diabetes mellitus (25.0%), cardiovascular disease (9.4%), stroke, arthritis, and osteoporosis.
OM is influenced by both environmental and host factors, and studies have investigated its prevalence in specific populations. The prevalence of OM confirmed either by inspection or mycological examination was investigated in specific populations, such as farmers, soldiers, psychiatric inpatients, and athletes. In addition, studies regarding occupations, such as sailors and navy cadets, with frequent exposure to water, which is considered a risk factor for OM, have also been conducted. This study is significant in that the subjects were district-based in addition to the relatively homogenous characteristics in the occupations. The prevalence of OM in individuals living in fishing villages located on two small islands in Korea is considered high. The prevalence of clinically diagnosed OM was 74.4%, and that of mycologically confirmed OM was 53.5%. These were much higher than those of other specific population groups: 16.0% in athletes, 17.7% in farmers, and 24.3% in psychiatric inpatients. In addition, the prevalence of OM among navy cadets was lower than our results. Identifying the number of actual residents was difficult. However, even with the number of administratively registered personnel (247 in two islands) that heavily outnumbered the actual target, the point prevalence was 13.0%, which was much higher than that in the general population in Korea (3.48~3.65%).
Furthermore, this study revealed that the degree of severity based on the number of involved nails and the percentage of the involved area was higher than that reported by a previous study involving the general population. In 62.5% of the study subjects, three or more nails were involved, whereas in 52.4% of the general population with OM, 1~2 nails were involved. The percentage of subjects with an involvement of more than 75% was higher in this study (40.6%) than that in the general population (32.5%). In addition, the rates of TDO (12.5% vs. 9.2~10.0%) and concomitant involvement of both the fingernail and toenail (12.5% vs. 3.4%) were higher in this study than those in the general population. Regarding the identified fungal strains, T. rubrum was the most common, followed by Trichophyton and Candida species, which was similar to the results reported by studies involving the general population.
It was assumed that the characteristic risk factors of OM in fishing villages are connected to the humid microenvironment, which might result from the use of occlusive equipment in addition to frequent exposure to water. It was likely that waterproof rubber materials could make the hand/foot easily sealed. This study showed increased ORs for gloves and rubber boots, but no statistical significance was observed. Wearing occlusive equipment alone did not significantly affect the prevalence of OM. However, compared with the hand, alleviating humidity of the foot during activity seemed to be more difficult, and the resulting sustained humidity might explain the increased ORs and the difference in the signifi- cance of toenail OM.
The prevalence of some commodities was higher in patients with OM in Korea. This may be attributed to the characteristics of fishing villages where the population is concentrated in elderly compared with the general population. In addition, this study showed that the rate of ongoing treatment for clinically diagnosed OM is low among individuals living in fishing villages. Even these treatments were not thought to be sufficient considering the mismatch be- tween most commonly used method and the increased severity of OM. Furthermore, the proportion of subjects who had arbitrarily discontinued the treatment was high. Inappropriate self-assessment and insufficient understanding of the disease or treatment process were the most common reasons for the discontinuation, whereas contrary to expectation, poor medical accessibility was hardly involved. It was assumed that the insufficient understanding of OM, which leads to a high rate of treatment discontinuation, along with the mismatch between the disease severity and the applied medications may have influenced the unfavorable status, in addition to the constitutive risk factor. In addition, it was revealed that subjects with OM tended to be more active in terms of behaviors for personal hygiene, such as taking a shower, washing the hands/feet, and drying. A study on the public awareness of OM has shown that 52.8% of subjects believed that washing and drying could cure OM, which is supported by our results. The limitation of this study was that not all residents were included. Selection bias might have also affected the results of this study. Considering the higher mean age of this study, old age seemed to have influenced the results.
This study suggests that the prevalence and severity of OM are higher in individuals living in fishing villages in Korea than those in the general population. Besides, most residents of fishing villages had a misbelief and low recognition of the OM treatment process. Therefore, more adequate and proper education with medical services for OM should be provided.
References
1. Craddock LN, Schieke SM. Superficial fungal infection: onychomycosis. In: Kang S, Amagai M, Bruckner AL, Enk AH, Margolis DJ, McMichael AJ, et al., editors. Fitzpatrick's dermatology, 9th ed. New York: McGraw-Hill, 2019:2937
2. James WD, Elston DM, Treat JR, Rosenbach MA, Neuhaus IM, editors. Andrew's diseases of the skin, 13th ed. Philadelphia: Elsevier, 2019:299
3. Lee JH, Han KD, Kim HJ, Han JH, Seo HM, Bang CH, et al. Prevalence of onychomycosis in Korea over 10 years (2006~2015). Korean J Dermatol 2018;56:655-657
Google Scholar
4. Lee JH, Lee HJ, Han KD, Seo HM, Bang CH, Lee JY, et al. Prevalence of onychomycosis in Korea: A nationwide population-based study. J Mycol Infect 2018;23:63-67
Google Scholar
5. Lipner SR, Scher RK. Onychomycosis: diagnosis and therapy. In: Razzaghi-Abyaneh M, Shams-Ghahfarokhi M, Rai M, eds. Medical Mycology: Current Trends and Future Prospects. Boca Raton, FL: CRC Press;2015:28
6. Scher RK, Rich P, Pariser D, Elewski B. The epidemiology, etiology, and pathophysiology of onychomycosis. Semin Cutan Med Surg 2013;32:S2-S4
Google Scholar
7. Papini M, Piraccini BM, Difonzo E, Brunoro A. Epidemi- ology of onychyomycosis in Italy: prevalence data and risk factor identification. Mycoses 2015;58:659-664
Google Scholar
8. Perea S, Ramos MJ, Garau M, Gonzalez A, Noriega AR, del Palacio A. Prevalence and risk factors of tinea unguium and tinea pedis in the general population in Spain. J Clin Microbiol 2000;38:3226-3230
Google Scholar
9. Sahin I, Kaya D, Parlak AH, Oksuz S, Behcet M. Dermato- phytoses in forestry workers and farmers. Mycoses 2005; 48:260-264
Google Scholar
10. Ingordo V, Fracchiolla S, Figliola F, D'Andria G, Colecchia B, Naldi L. Prevalence and awareness of tinea pedis in Italian sailors. Dermatology 2000;201:349-350
Google Scholar
11. Ingordo V, Naldi L, Fracchiolla S, Colecchia B. Prevalence and risk factors for superficial fungal infections among Italian navy cadets. Dermatology 2004;209:190-196
Google Scholar
12. Djeridane A, Djeridane Y, Ammar-Khodja A. A clinico- mycological study of fungal foot infections among Algerian military personnel. Clin Exp Dermatol 2007;32: 60-63
Google Scholar
13. Kawai M, Suzuki T, Hiruma M, Ikeda S. A retrospective cohort study of tinea pedis and tinea unguium in in- patients in a psychiatric hospital. Med Mycol J 2014; 5:E35-41
Google Scholar
14. Sabadin CS, Benvegnu SA, da Fontoura MM, Saggin LM, Tomimori J, Fischman O. Onychomycosis and tinea pedis in athletes from the state of Rio Grande Do Sul (Brazil): a cross-sectional study. Mycopathologia 2011;171:183-189
Google Scholar
15. Hwang SM, Kim DM, Suh MK, Kwon KS, Kim KH, Ro BI, et al. Epidemiologic survey of onychomycosis in Koreans: multicenter study. Korean J Med Mycol 2011;16:35-43
Google Scholar
16. Gupta AK, Jain HC, Lynde CW, MacDonald P, Cooper EA, Summerbell RC. Prevalence and epidemiology of onycho- mycosis in patients visiting physicians' offices: a multi-center Canadian survey of 15,000 patients. J Am Acad Dermatol 2000;43:244-248
Google Scholar
17. Piraccini BM, Alessandrini A. Onychomycosis: A review. J Fungi 2015;1:30-43
Google Scholar
18. Ahn HS, Park HJ, Lee JY, Cho BK. Classification of onycho- mycosis in Korea. Korean J Med Mycol 2008;13:129-137
Google Scholar
19. Lim SW, Suh MK, Ha GY. Clinical features and identifi- cation of etiologic agents in onychomycosis (1999-2002). Korean J Dermatol 2004;42:53-60
Google Scholar
20. Roh D, Kim H, Nam JH, Mun JH, Park J, Park J, et al. Public awareness of onychomycosis in Korean: A survey of 621 participants. J Mycol Infect 2018;23:15-23
Google Scholar
Congratulatory MessageClick here!